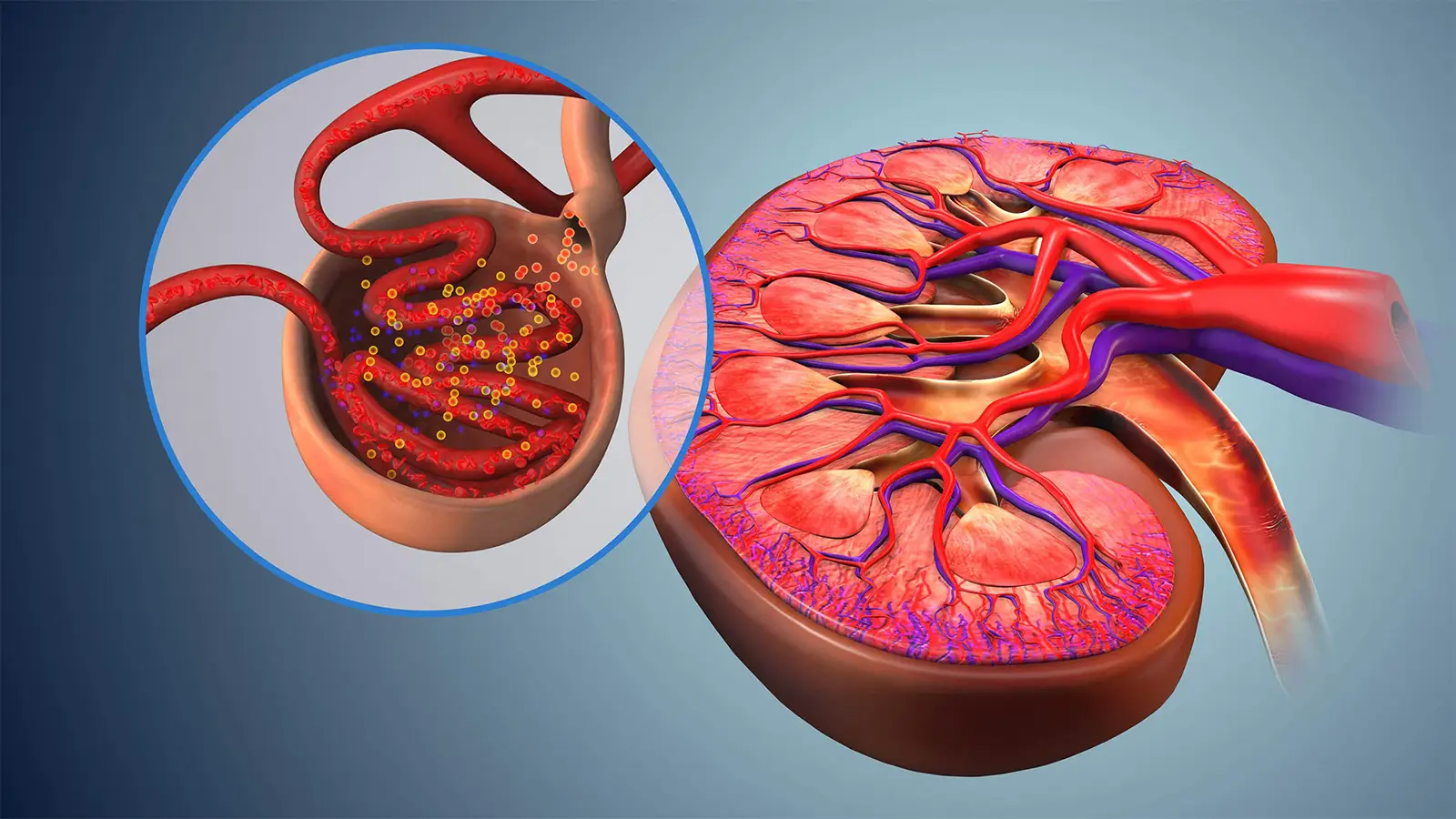
IgA nephropathy is a rare condition caused by excessive buildup of immunoglobulin A in the kidneys, slowly but progressively degrading renal function to the point of requiring dialysis or transplantation.Treatment has historically focused on manifestations such as high blood pressure, but more targeted therapies have been introduced in recent years.This phase III trial with telitacicept, a dual-action biologic drug that inhibits B-cell activating factor and the so-called APRIL protein, showed that it outperformed placebo in reducing proteinuria in a 39-week interim analysis.
Patients with immunoglobulin A (IgA) nephropathy had less protein in their urine -- a key surrogate marker for the renal impairment caused by the condition -- after treatment with a dual-acting biologic agent that inhibits two regulatory cytokines, researchers said.
Urinary protein-to-creatinine ratio fell by 58.9% after 39 weeks of therapy with telitacicept, a drug already approved in China for lupus, compared with an 8.8% decline in those treated with placebo injections, according to Hong Zhang, MD, PhD, of Peking University First Hospital in Beijing, and colleagues.
Telitacicept also appeared to be safe. Few adverse events tracked in the study were substantially more common with the drug versus placebo; the only exceptions were injection site reactions and some diminution in titers of other immunoglobulins, the researchers reported in the New England Journal of Medicine.