Fierce Pharma
Fierce Biotech
Fierce Healthcare
Fierce Life Sciences Events
Advertise
AstraZeneca’s first foray with PD-L1 inhibitor Imfinzi as a treatment for bladder cancer was a flop. | Following an FDA approval in muscle-invasive bladder cancer patients who are eligible to receive cisplatin-based chemotherapy, AstraZeneca on Thursday reported positive phase 3 results for its Imfinzi, used alongside Padcev, in those who were cisplatin-ineligible.
Fierce Pharma
Fierce Biotech
Fierce Healthcare
Fierce Life Sciences Events
Advertise
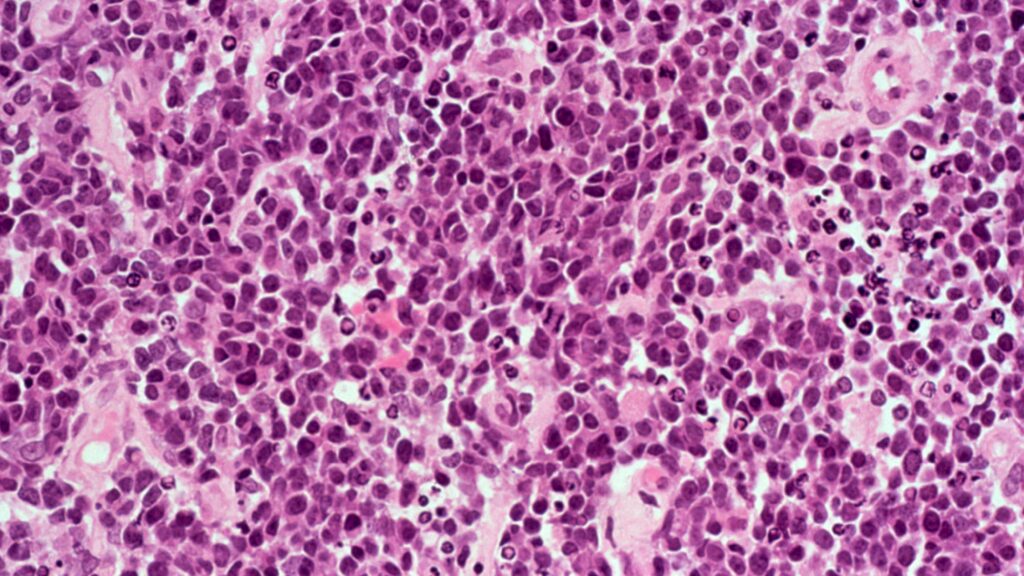
BeOne wins FDA approval in lymphoma race

FDA blocked melanoma drug as confusion reigned under Makary

Astellas treatment offers new hope to a devastated rare disease community
Tumori, una nuova immunoterapia disponibile per tre neoplasie
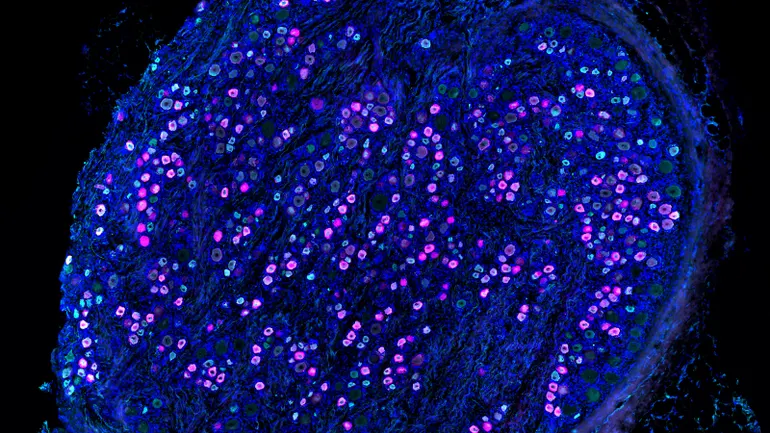
A year after Vertex’s big launch, pain drug research faces a pivotal moment

The advisory panel wasn't convinced that a clinical trial proved that early switching to camizestrant improved long-term survival…

The experimental drug reduced flare-ups for patients with chronic obstructive pulmonary disease, Astra said Friday.

Pierre Fabre claims to now be “aligned” with U.S. regulators on the path forward for a twice-rejected treatment. Elsewhere,…
The F.D.A. approved Merck’s injected version of its blockbuster infusion Keytruda. The company says it will be quicker and…
The FDA is looking to get thrifty with its approved drugs. | The framework would leverage existing safety data on approved drugs…

Si amplia la gamma di pazienti oncologici che possono beneficiare del trattamento con l'immunoterapia. L'Agenzia Italiana del…