The approval covers all 27 EU member states, Liechtenstein, Norway, and Iceland.
The approval is based on Phase III BASIS trial data showing Hympavzi achieved a 93% reduction in bleeding rate. Credit: PaeGAG / Shutterstock.com.
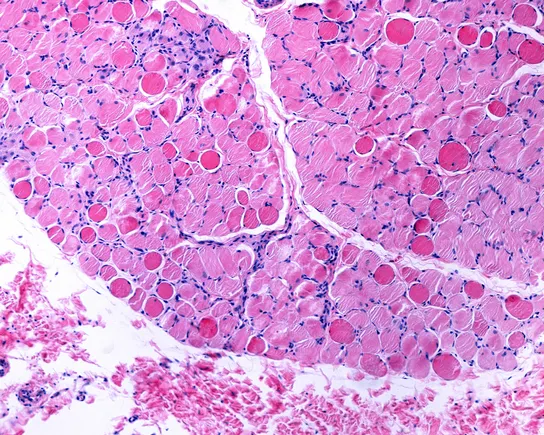
Pfizer has received the European Commission’s (EC) marketing authorisation for expanding the approved Hympavzi (marstacimab) indication to treat haemophilia A or B with inhibitors in patients aged 12 years and above.
The approval covers adults and adolescents in the European Union (EU) who weigh at least 35kg and have haemophilia A (factor VIII deficiency) with factor VIII inhibitors, or haemophilia B (factor IX deficiency) with factor IX inhibitors. Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.