Following the update, healthcare providers can increase both the dose and frequency of Crysvita for XLH patients after treatment.
The recombinant, fully human monoclonal IgG1 antibody targets fibroblast growth factor 23 in XLH. Credit: Michele Ursi / Shutterstock.com.
Kyowa Kirin has received approval from the US Food and Drug Administration (FDA) for a dosing update to the prescribing information for Crysvita (burosumab-twza), introducing a new option for adults with X-linked hypophosphataemia (XLH).
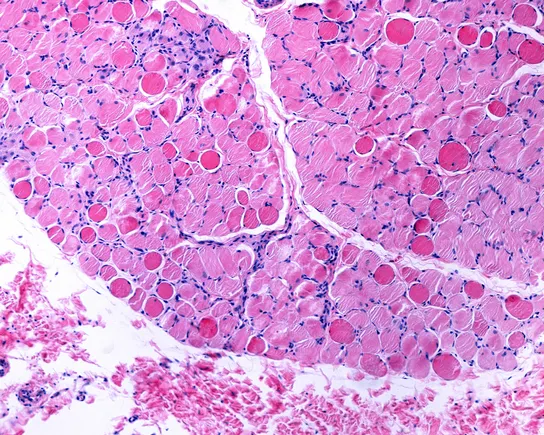
Affecting adults and children, this rare, progressive, genetic disorder impacts both bone and muscle health due to the body’s inability to retain sufficient phosphorus, a mineral essential for bone health. Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.