In this article
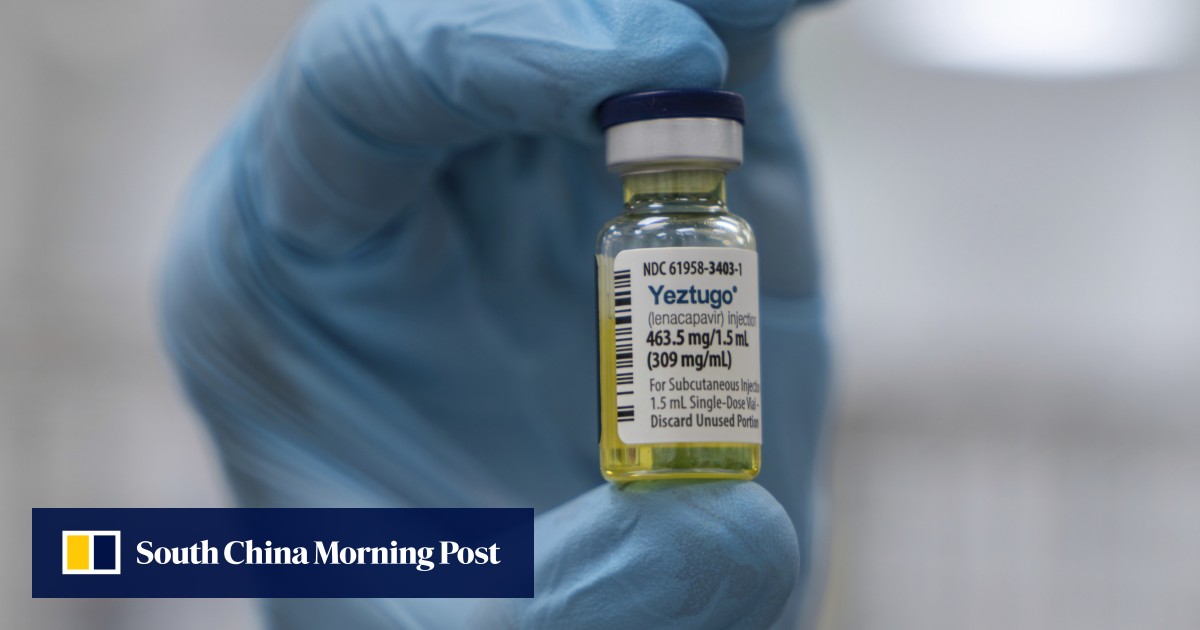
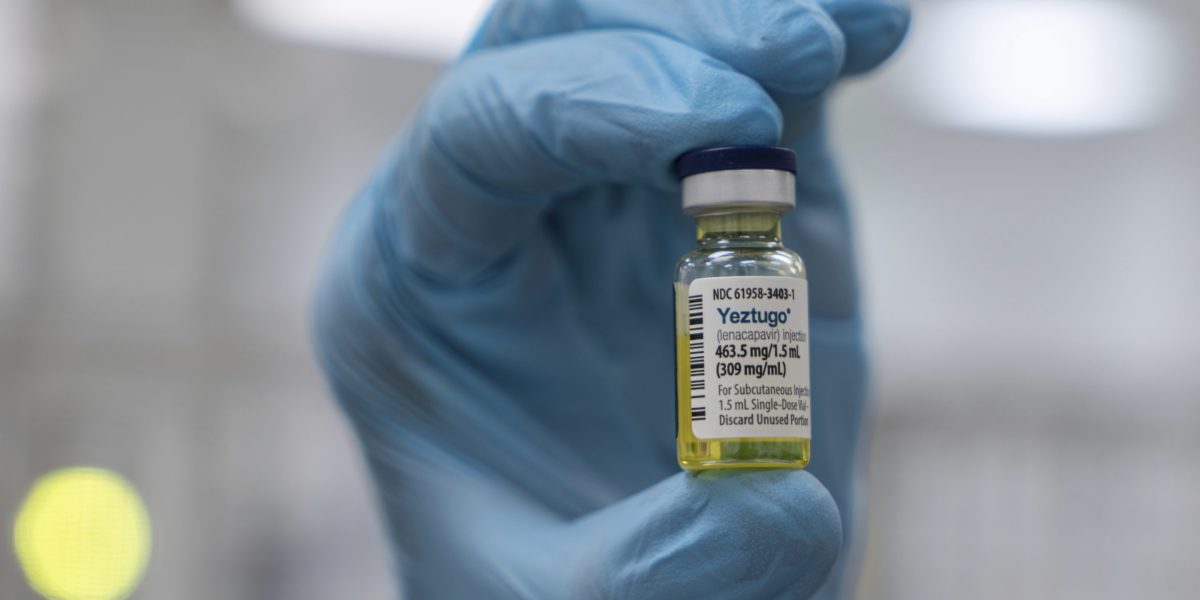
The Food and Drug Administration on Wednesday approved Gilead’
s twice-yearly antiviral injection for preventing HIV – a milestone that the company and some experts say could help bring the world closer to ending the decades-long epidemic caused by the virus.
But the launch of the injectable drug, which will be marketed under the name Yeztugo, faces a set of potential threats, including the Trump administration’s proposed cuts to federal funding for HIV prevention efforts.
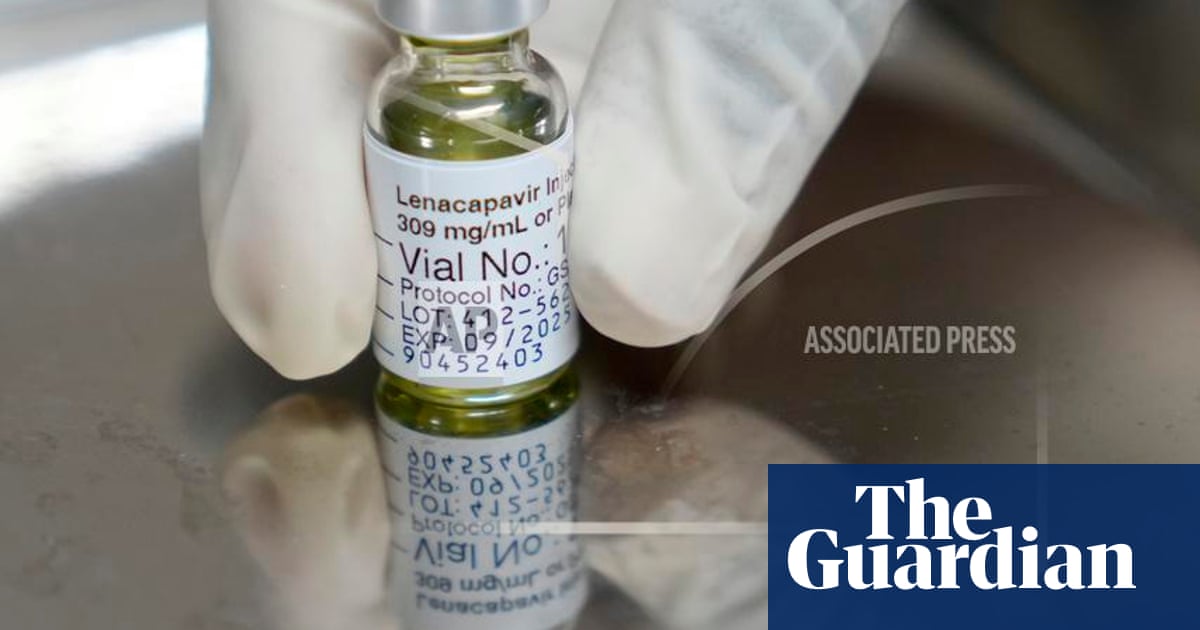
In two groundbreaking clinical trials last year, Gilead’s injection proved to be capable of virtually eliminating new HIV infections when taken every six months. Patients take it less frequently than all existing HIV prevention medications, including daily pills from Gilead and another injection from GSK