Home
Storia in 7 fonti
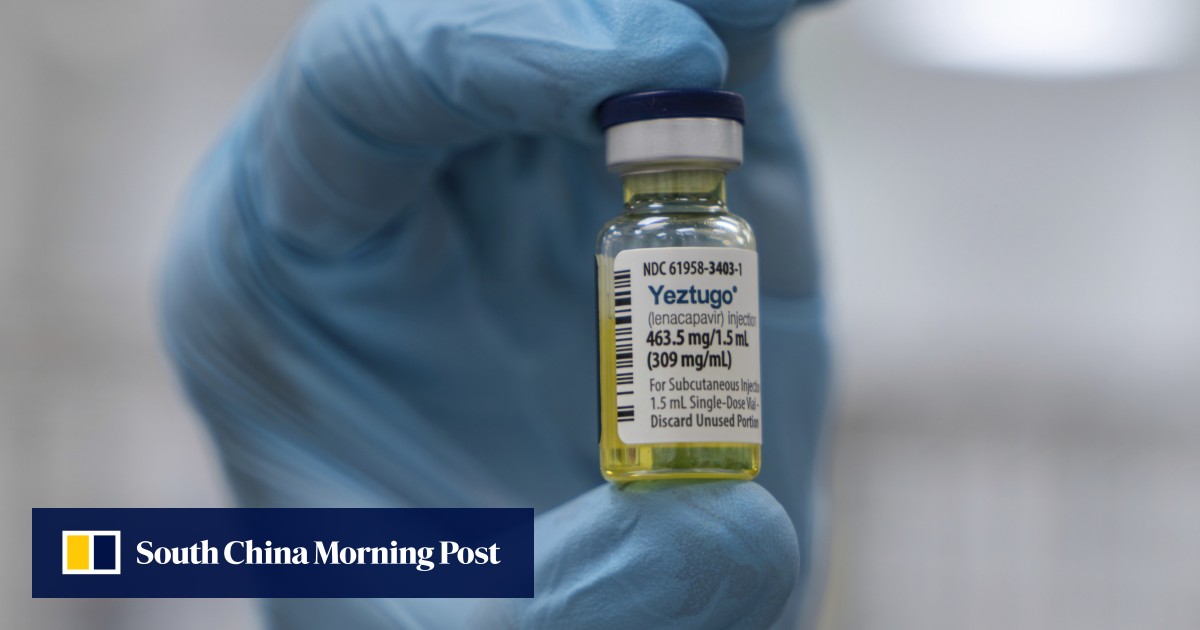
FDA approves Gilead's twice-yearly HIV prevention injection, offering a powerful and convenient new option
The launch of the injection faces potential threats, including the Trump administration's proposed cuts to federal funding for HIV prevention efforts.
- ·
usatoday.com

FDA approves new twice-yearly HIV shot. What to know
The FDA approved Gilead's HIV prevention drug lenacapavir, a twice-a-year injectable medication that clinical trials show prevents new infections.
- ·
cnbc.com

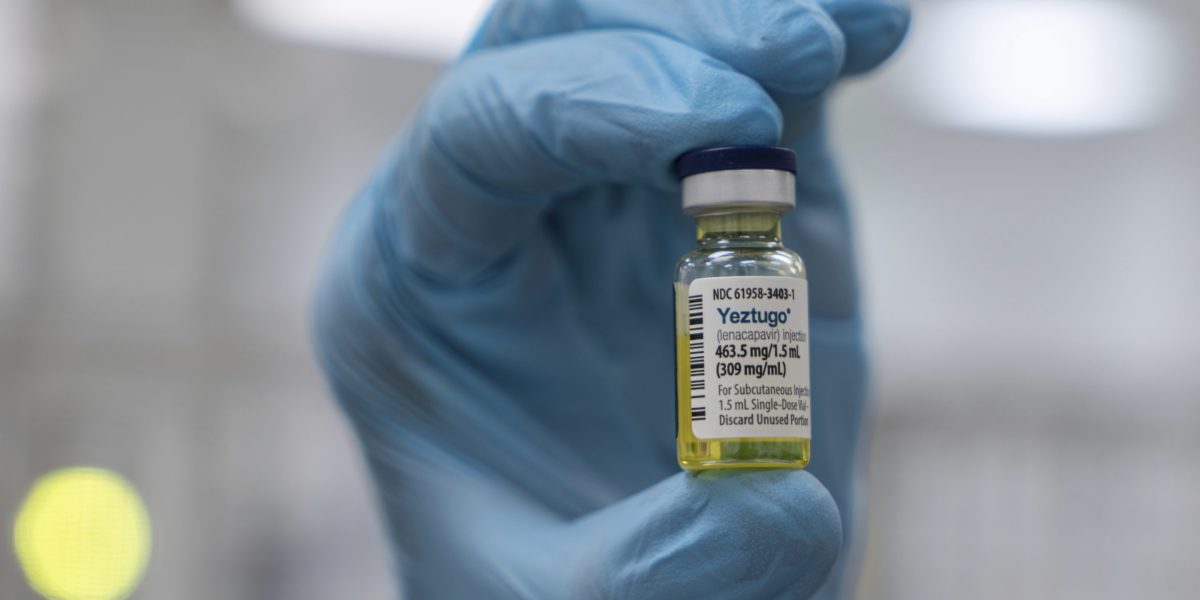
FDA approves Gilead's twice-yearly HIV prevention injection, offering a powerful and convenient new option
The launch of the injection faces potential threats, including the Trump administration's proposed cuts to federal funding for HIV prevention efforts.