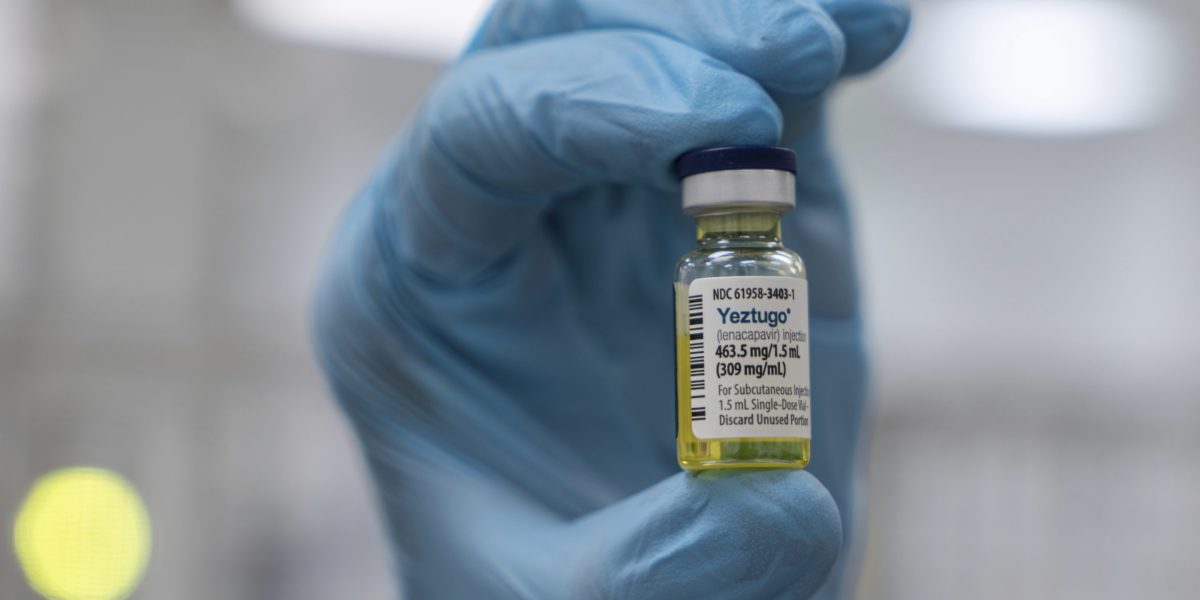
The Food and Drug Administration on Wednesday approved Gilead's HIV prevention drug lenacapavir, a twice-a-year injectable medication that clinical trials show prevents new infections.
Gilead did not immediately say how much it will charge for the biannual shot, but company officials said it would be comparable to existing HIV prevention drugs.
The drug will be marketed under the name Yeztugo.
Advocates say the long-acting medication is promising because it's more convenient than existing HIV prevention drugs that must be taken daily.
Giving people the option of a twice-a-year injectable is "a game changer, not only for the United States, but around the world," said Carl Schmid, executive director of the HIV+Hepatitis Policy Institute.