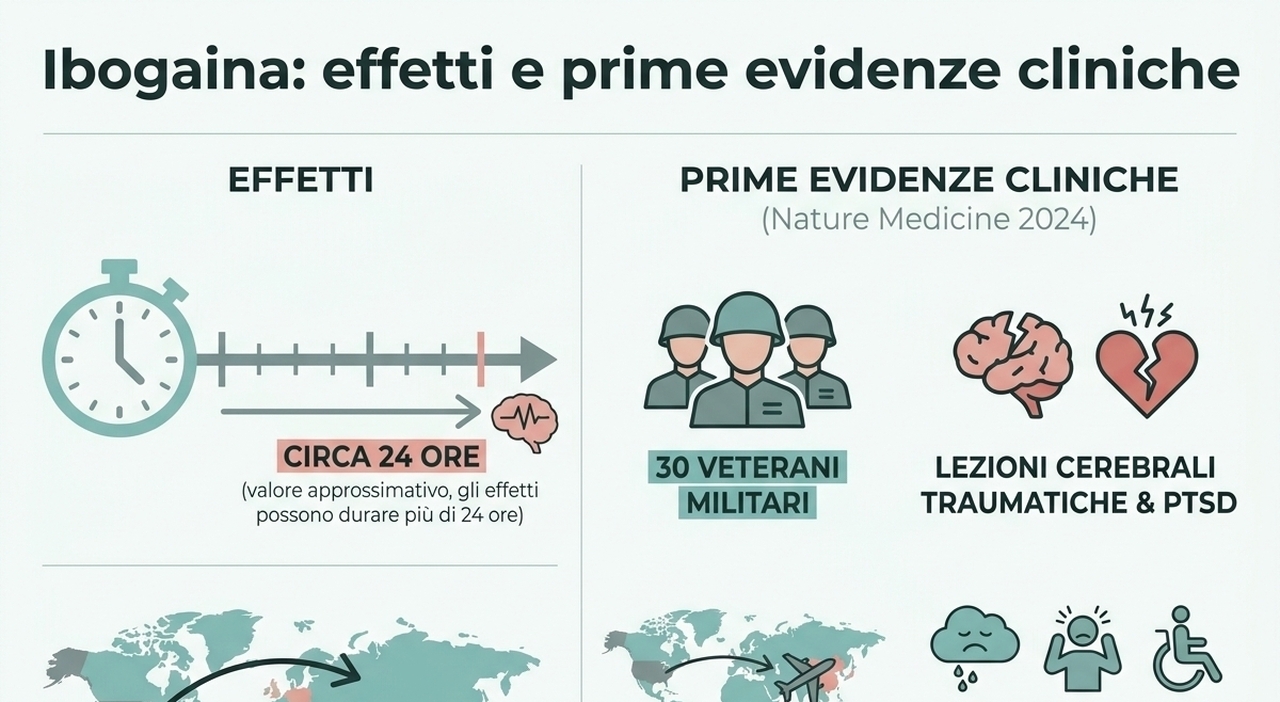
April 24 (UPI) -- The Food and Drug Administration announced regulatory actions Friday to support development of serotonin-2A agonists and related products, including psychedelic drugs.
On Saturday, President Donald Trump signed an executive order to speed up research on psychedelic drugs for mental health disorders.
"Today's order will ensure that people suffering from debilitating symptoms might finally have a chance to reclaim their lives and lead a happier life," Trump said during the signing.
Friday, the FDA said it is issuing national priority vouchers to three companies studying: psilocybin for treatment-resistant depression and major depressive disorder; and methylone for post-traumatic stress disorder.
Related