The U.S. Food and Drug Administration on Friday announced a series of measures aimed at accelerating the development of psychedelic treatments for serious mental illness.
That comes after President Donald Trump signed an executive order on Saturday directing federal health agencies to expand access to emerging therapies.
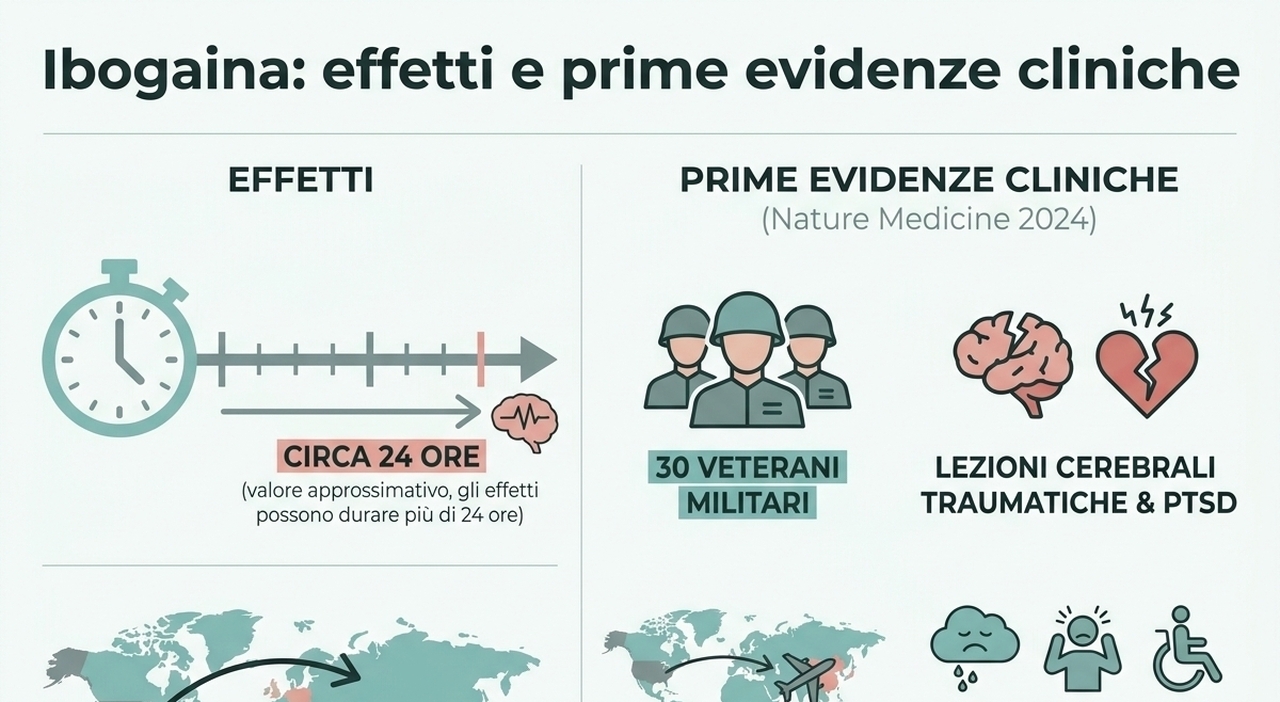
The move marks a significant shift toward supporting psychedelic-based medicines for conditions such as treatment-resistant depression, post-traumatic stress disorder and other substance use disorders, the FDA said.
“Under President Trump’s leadership, we are accelerating the research, approval and responsible access to promising mental health treatments,” Robert F. Kennedy Jr., secretary of the U.S. Department of Health and Human Services, said in the release. “The FDA will prioritize therapies with Breakthrough Therapy designation, where early evidence shows meaningful improvement.”
As part of the announcement, the FDA said it would issue national priority vouchers to companies studying psilocybin for depression and methylone for PTSD.