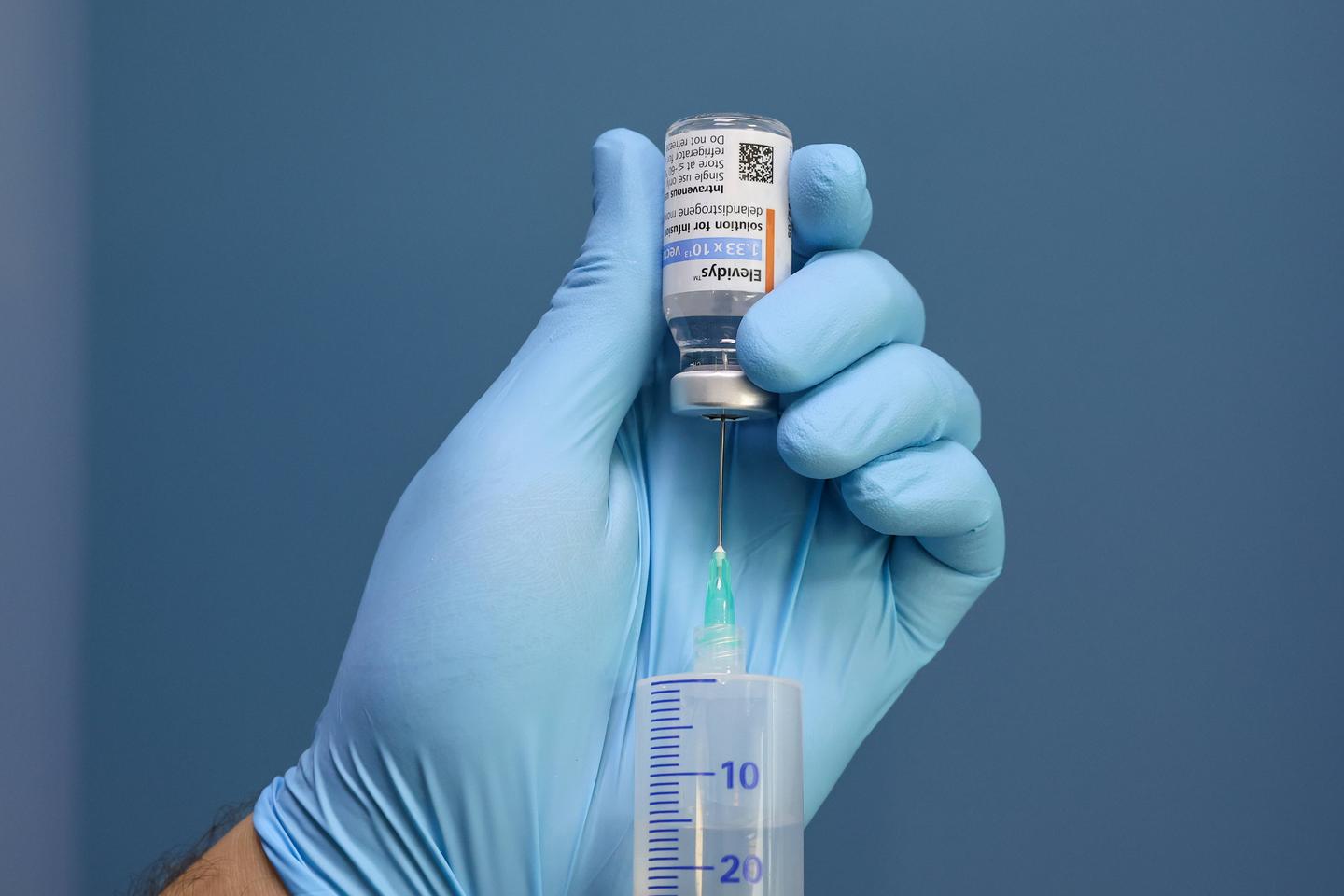
The drug, which was designed to treat Duchenne muscular dystrophy, has been linked to the deaths of two teenage boys.
It has been a grim few months for the Duchenne muscular dystrophy (DMD) community. There had been some excitement when, a couple of years ago, a gene therapy for the disorder was approved by the US Food and Drug Administration for the first time. That drug, Elevidys, has now been implicated in the deaths of two teenage boys.
The drug’s approval was always controversial—there was a lack of evidence that it actually worked, for starters. But the agency that once rubber-stamped the drug has now turned on its manufacturer, Sarepta Therapeutics. In a remarkable chain of events, the FDA asked the company to stop shipping the drug on July 18. Sarepta refused to comply.
For many patients, pushing for access to unproven treatments is their best chance of survival. And that’s worth the risk.
In the days since, the company has acquiesced. But its reputation has already been hit. And the events have dealt a devastating blow to people desperate for treatments that might help them, their children, or other family members with DMD.