Fierce Pharma
Fierce Biotech
Fierce Healthcare
Fierce Life Sciences Events
Advertise
With the help of DNA testing company Natera and its personalized molecular residual disease (MRD) blood test Signatera, Roche’s PD-L1 inhibitor Tecentriq has chalked up its eleventh U.S. | The FDA approved the Tecentriq label expansion alongside Natera's Signatera as a companion diagnostic.
Fierce Pharma
Fierce Biotech
Fierce Healthcare
Fierce Life Sciences Events
Advertise
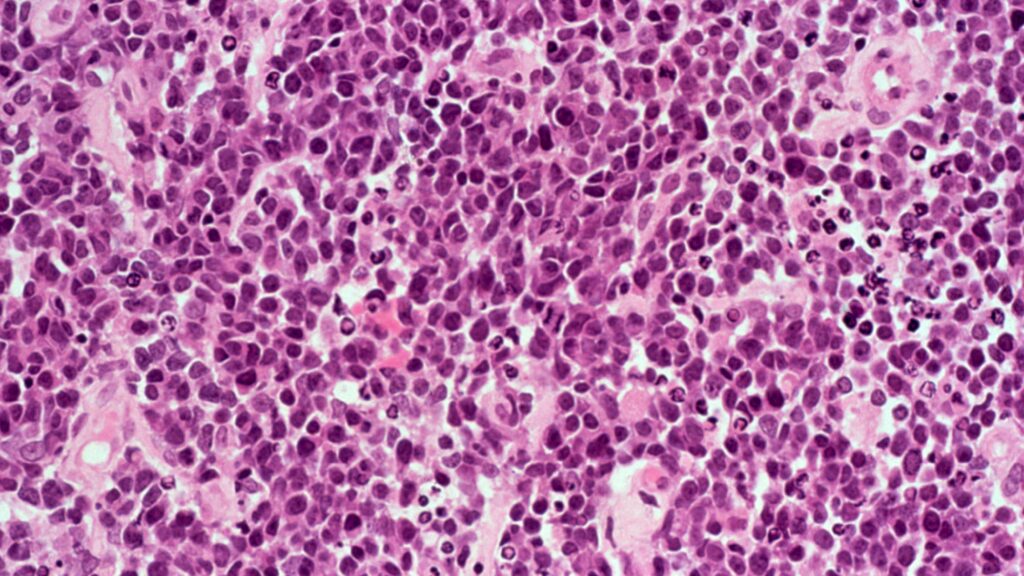
BeOne wins FDA approval in lymphoma race

Roche Pharma launches India’s first 7-minute injectable immunotherapy for lung cancer

Advancing Frontline Care With a Quadruplet Regimen for Transplant-Ineligible Myeloma Patients

Updated: BeOne’s next-gen BCL2 inhibitor wins FDA approval, taking aim at Venclexta

A Series A, B and an IPO; Cabaletta reports CAR-T results

AstraZeneca follows Merck with Phase 3 win in bladder cancer

Inhibrx delivers ‘differentiated’ cancer drug data; Fractyl starts diabetes gene therapy trial
Arvinas, Pfizer find new partner for ‘Protac’ breast cancer drug
CREATE Medicines, a biotech company developing CAR-T therapies, raises $122 million
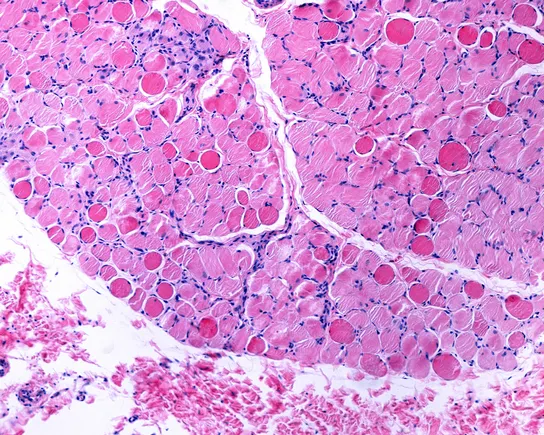
With new data, Regenxbio to seek FDA approval of Duchenne gene therapy