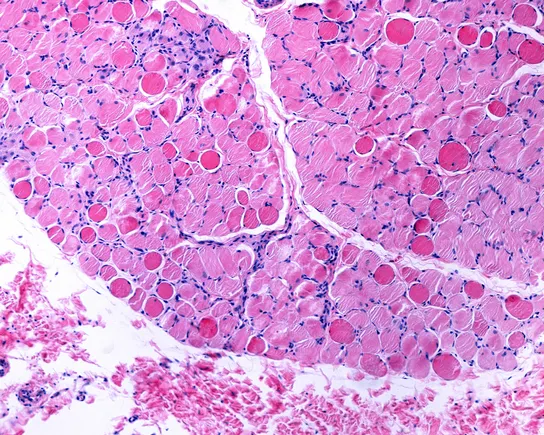
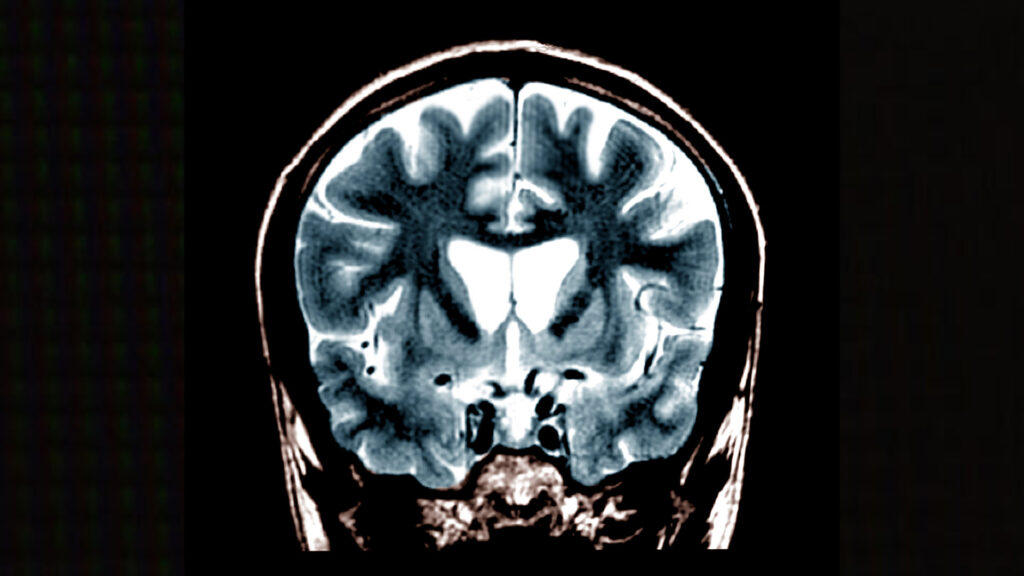
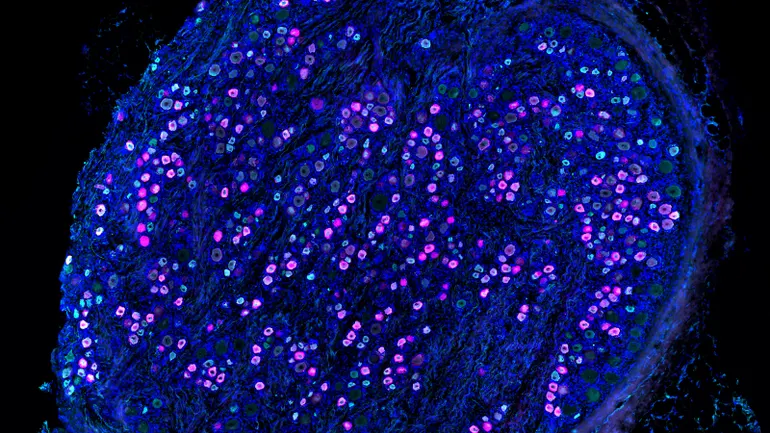
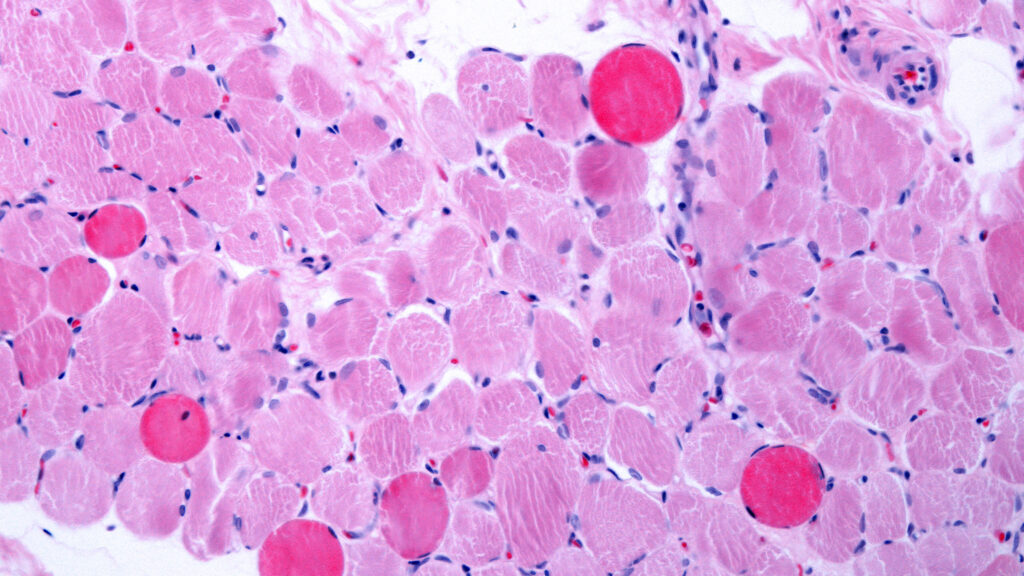
Developing a new drug can cost a billion dollars and take more than a decade. That makes investing in new treatments in the rare disease space — where patient populations are small, and the chance of earning a return on that investment even smaller — a risky bet for big pharmaceutical companies.
That’s a big reason why 95% of the more than 10,000 rare diseases that exist do not have an FDA-approved treatment.
But for parents of children diagnosed with those diseases, doing nothing is not an option. And that’s fueling a major trend in the rare disease community: medical innovation and breakthroughs that are being driven by the patient groups themselves.
Last October, Rare As One — a project funded by the Chan Zuckerberg Biohub that supports patient-led research in the rare disease space — released a report that showed of the 20 organizations it funded when the program launched in 2019, half of them were involved in clinical trials within five years.
That’s remarkable progress from advocacy groups that are mostly led by the communities they represent – regular parents from varied backgrounds, partnering with researchers and clinicians to develop life saving treatments for their loved ones. But for many of these families, figuring out where to start that process can be incredibly complicated.