Hyderabad-based paediatrician Dr Sivarajanini Santosh during a virtual press conference on April 1 defended her campaign against certain electrolyte drinks. Credit: Screengrab
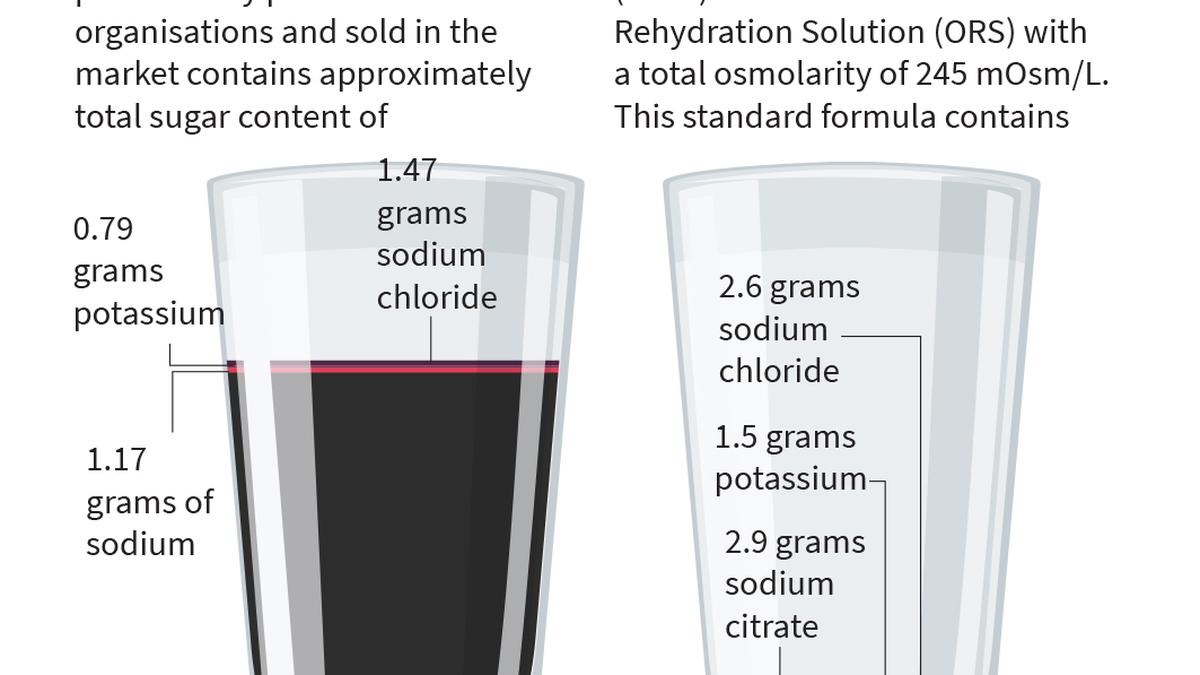
Hyderabad-based paediatrician Dr. Sivaranjani Santosh on Wednesday (April 1) defended her campaign against certain electrolyte drinks, alleging that misleading branding and high sugar content in products such as ORSL and its rebranded version ERZL pose a risk to children’s health.Addressing a virtual press conference, she responded to a legal notice issued by Johnson & Johnson and Kenvue, accusing the companies of attempting to intimidate her.The pharmaceutical major issued a notice to Dr. Santosh on March 16, accusing her of making false, misleading, disparaging and defamatory statements about their products.Setting the context, Dr. Santosh highlighted the scale of the public health issue, noting that diarrhoea kills between 4.5 lakh and 5.5 lakh children globally each year, including around 1 lakh to 1.5 lakh in India, making it the third leading cause of death among children under five years. She explained that Oral Rehydration Salts (ORS) are scientifically formulated to rehydrate effectively, with a reduced osmolarity solution of 245 mOsm/L shown to reduce the need for IV fluids by nearly 40%, lower stool output by 25–30%, and decrease vomiting.She contrasted this with high-sugar electrolyte drinks available in the Indian market, stating that excessive sugar increases osmolarity and draws water into the gut, worsening diarrhoea.Tracing the history of ORSL, she said the product was launched in 2003 and later acquired by Johnson & Johnson in 2014, after which its marketing intensified. She alleged that the product was widely sold as a substitute for ORS, leading to confusion among parents.“Despite objections from paediatricians and even a legal case filed by a parent, the branding continued, with only a small disclaimer stating it should not be used during diarrhoea,” she said.Dr. Santosh filed a Public Interest Litigation (PIL) in 2022 and said organisations such as the Endocrine Society of India and the Women Paediatricians Forum joined her.“We also collected evidence from parents and doctors, including cases where children’s conditions worsened after being given ORSL instead of ORS. In one instance, a child with diabetes developed diabetic ketoacidosis after being given multiple packs of ORSL during diarrhoea,” she said.Following regulatory action in October 2025, she noted that authorities reiterated that ORS branding could not be used for non-compliant products. However, the company reintroduced the product as ERZL with minimal changes, retaining similar packaging and branding, thereby continuing to mislead consumers. She also objected to the practice of re-labelling existing ORSL stock and using the same branding for a new ‘low sugar’ variant containing sucralose.Calling this a violation of both regulatory orders and Delhi High Court directions, Dr. Santosh said the continued marketing of ERZL as a successor to ORSL created confusion, particularly among less aware consumers in smaller towns and rural areas.Dr Santosh outlined her demands, including an unconditional public apology from the companies to affected families, complete rebranding of the product to eliminate confusion, and a halt to marketing such drinks as daily hydration options for children. She also called on the government to strengthen regulations, improve coordination between regulatory bodies such as CDSCO and FSSAI, and restrict the sale of non-ORS liquids in pharmacies.Concluding her remarks, Dr. Santosh said her campaign was driven solely by concern for children’s health and warned that silencing such voices would discourage others from speaking out on public health issues. Published - April 01, 2026 04:03 pm IST