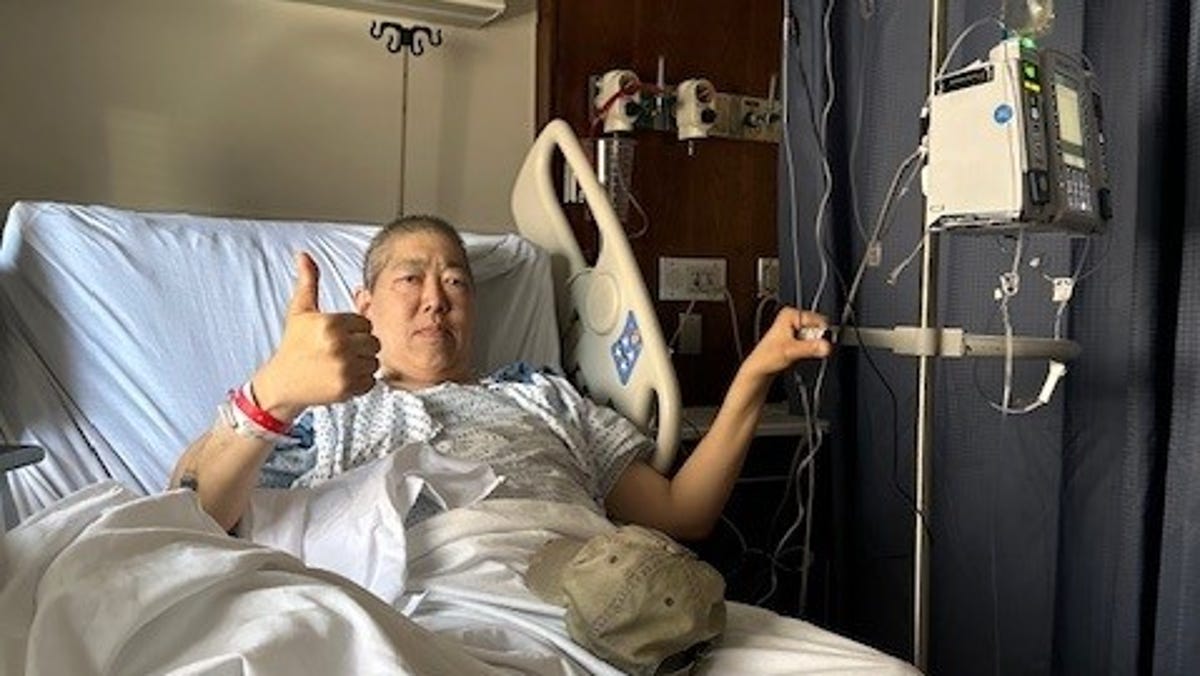
Jeff Smith was dumbfounded when he received a colon cancer screening kit in the mail.
The 68-year-old Minnesota man hadn't asked for the Cologuard test, and his doctor hadn't mentioned anything about it. And why in the world would he need such a test? Smith has lived most of his life without a colon. As a child, he had ulcerative colitis, an inflammatory bowel disease. Doctors removed his colon.
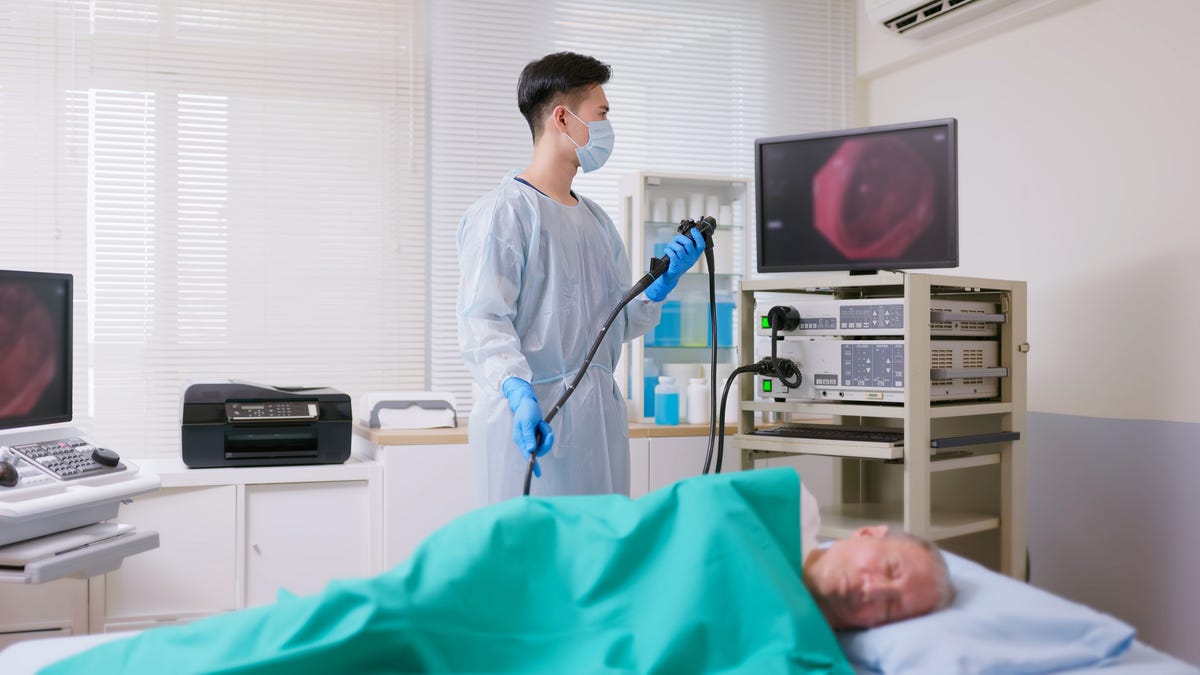
Smith's experience, which included additional uncomfortable testing after he completed the kit, has some doctors worried about the idea of mailing tests to patients without consulting their doctors first. Early detection of colon cancer is crucial – it's the second-leading cause of cancer deaths in the United States, with nearly 53,000 people projected to die from it in 2025, according to the American Cancer Society.
But the disease is highly treatable if caught early; screening is recommended for adults ages 45 to 75 at average risk. Yet nearly 1 in 3 eligible adults are not up to date on colon cancer screening.
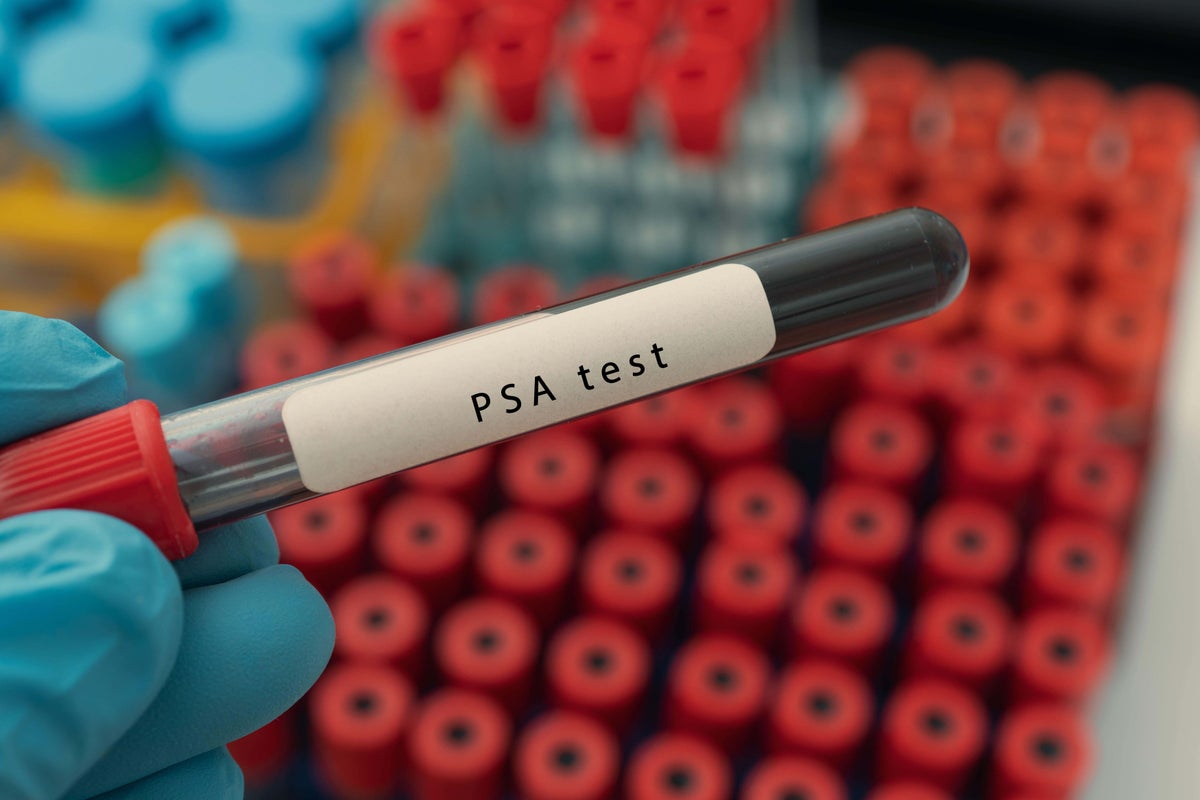
Consumers increasingly have testing options beyond the once-a-decade colonoscopy. Tests that can detect signs of the disease in a person's stool are available, and the Food and Drug Administration approved a blood test in 2024. Exact Sciences, the company that markets the Cologuard test, teamed up with health insurance companies and health systems to send kits to the homes of eligible people who have never been screened for colon cancer or are past due.