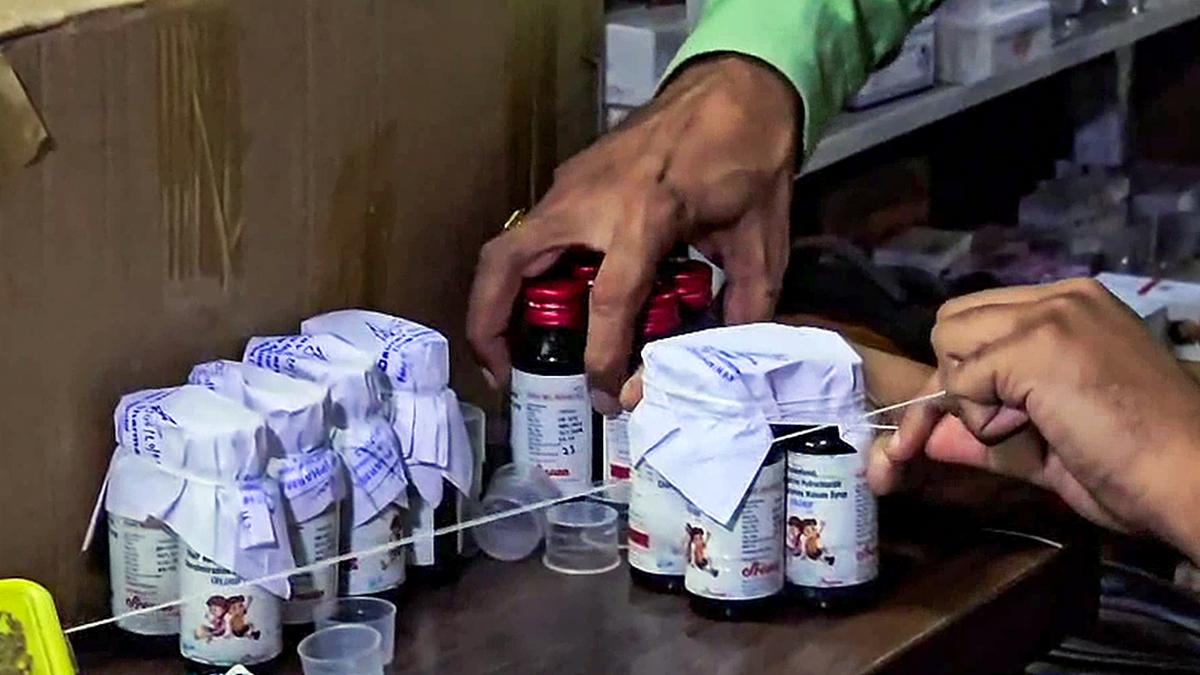
The Maharashtra Food and Drugs Administration (FDA) on Sunday (October 5, 2025), issued an urgent advisory calling for the immediate suspension of sale, distribution, and use of Coldrif Syrup, following reports of child fatalities in Madhya Pradesh and Rajasthan allegedly linked to the product.
Uttar Pradesh bans Coldrif cough syrup after 14 deaths in Madhya Pradesh
The affected batch, Coldrif Syrup (Phenylephrine Hydrochloride, Chlorpheniramine Maleate), Batch No. SR-13, was manufactured in May 2025 by Sresan Pharma, based in Sunguvarchathiram, Kancheepuram district, Tamil Nadu. It is set to expire in April 2027. Laboratory analysis revealed the presence of Diethylene Glycol (DEG), a toxic chemical commonly found in industrial products like antifreeze. DEG ingestion can cause kidney failure and has been linked to multiple deaths.
Speaking to The Hindu, FDA Drug Controller D.R. Gahane said, “Our State and citizens are safe. We held a meeting with the Drug Control Authorities in Tamil Nadu to trace the distribution of the batch in Maharashtra and they have confirmed that Coldrif cough syrups were not supplied in Maharashtra. It is difficult to trace now if someone had travelled from Maharashtra to Madhya Pradesh or other locations and coincidentally if they bought the cough syrup and brought it here. Still for safety, we had decided to issue the statement for public interest and safety.”