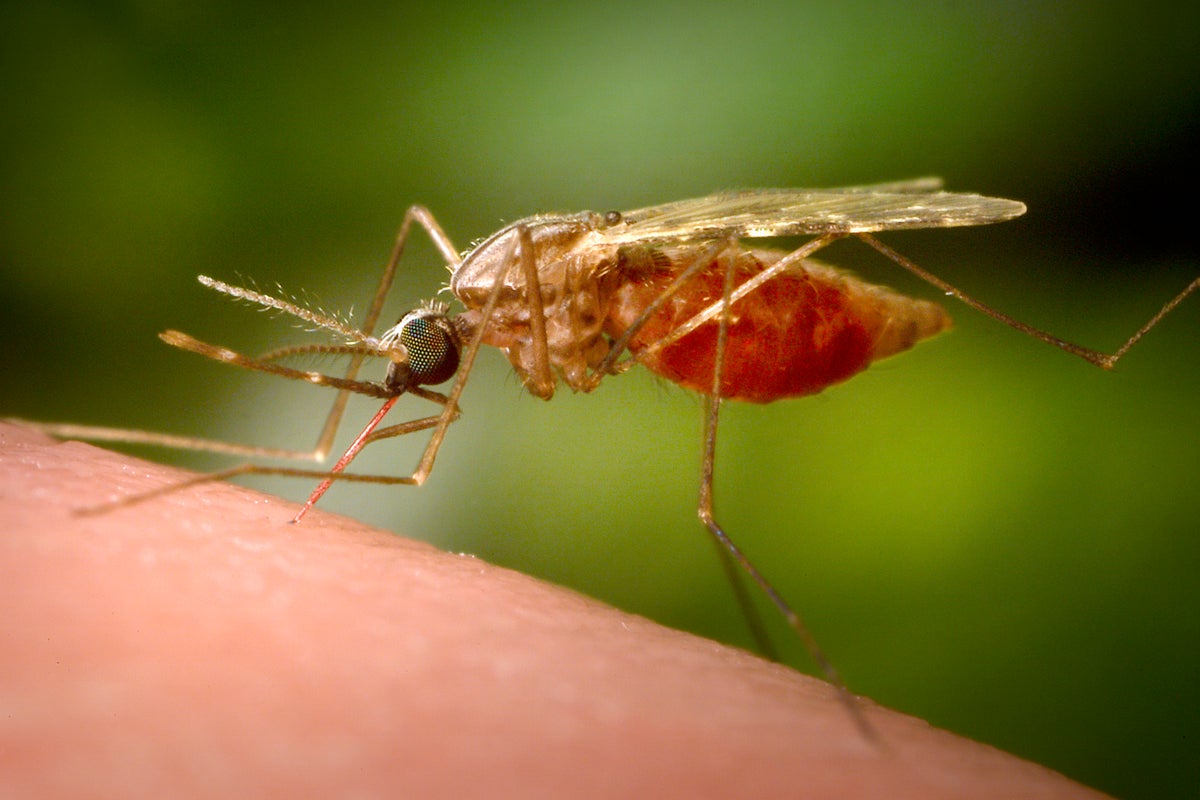
The decision to suspend the Ixchiq vaccine comes as public health experts warn the mosquito-spread virus could pose a future pandemic threat
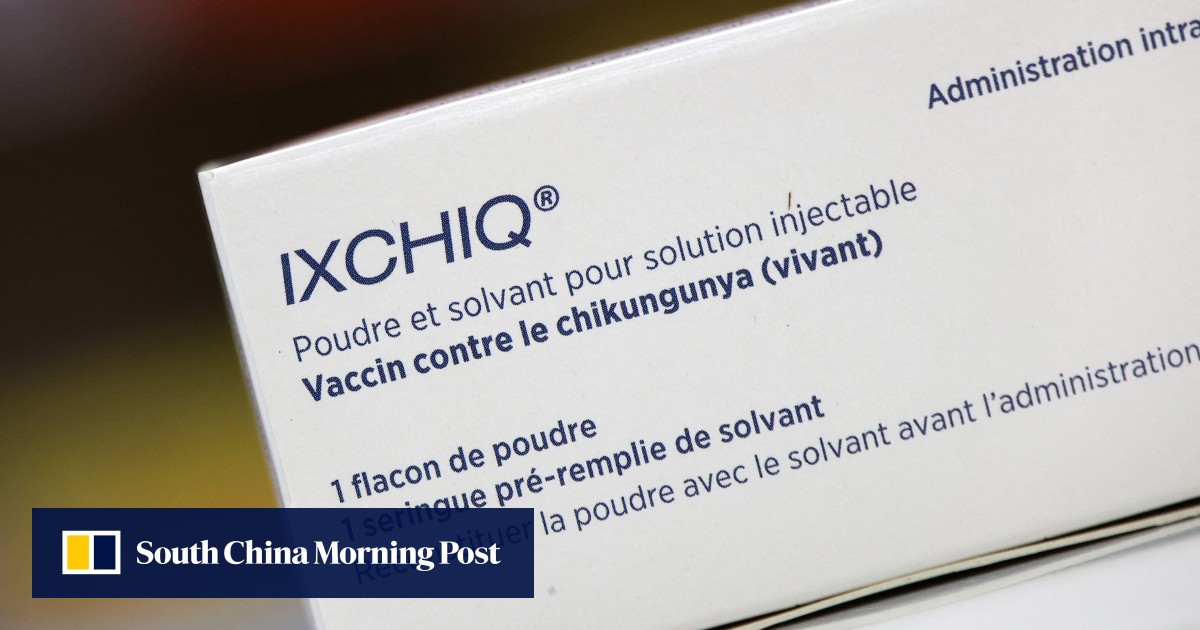
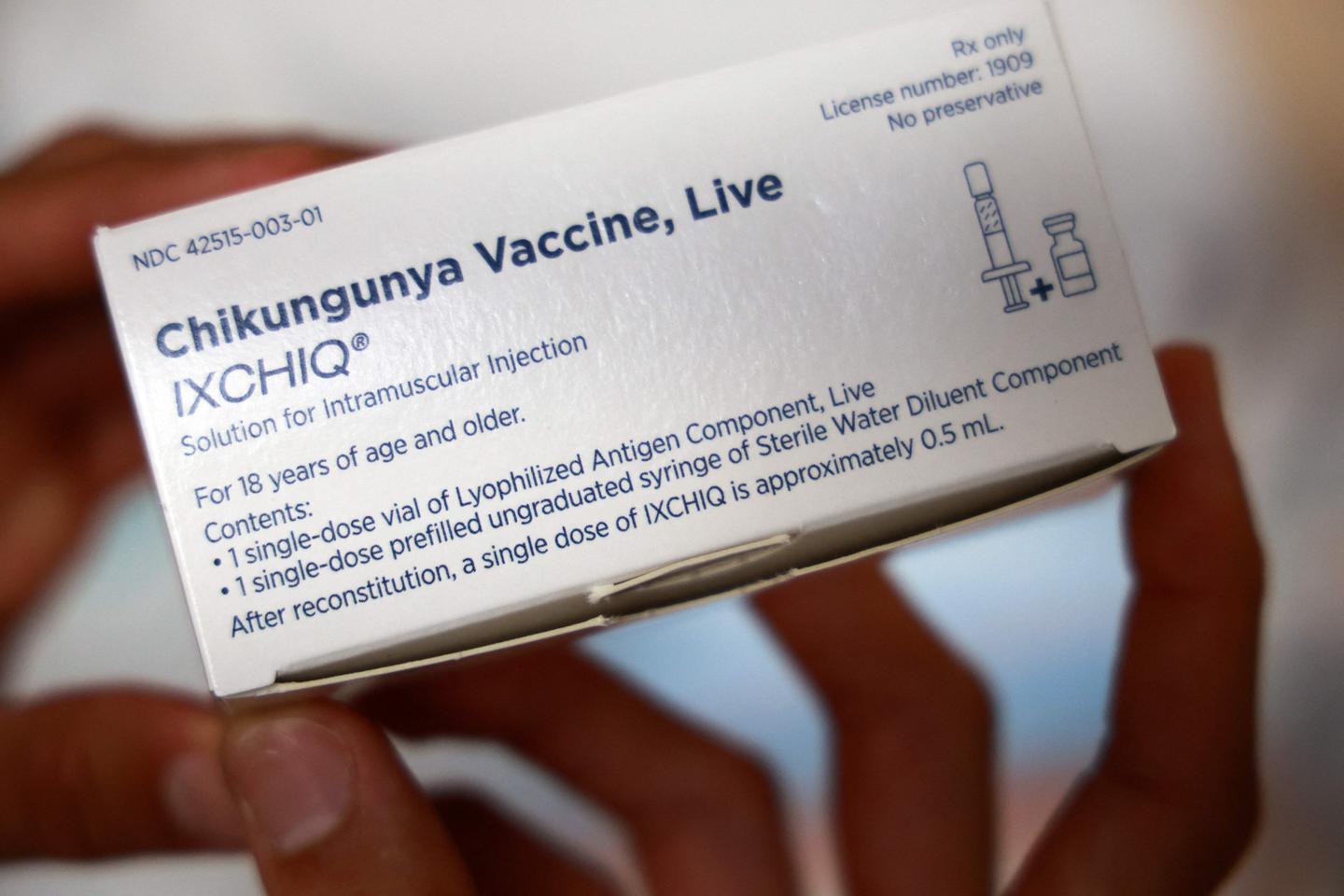
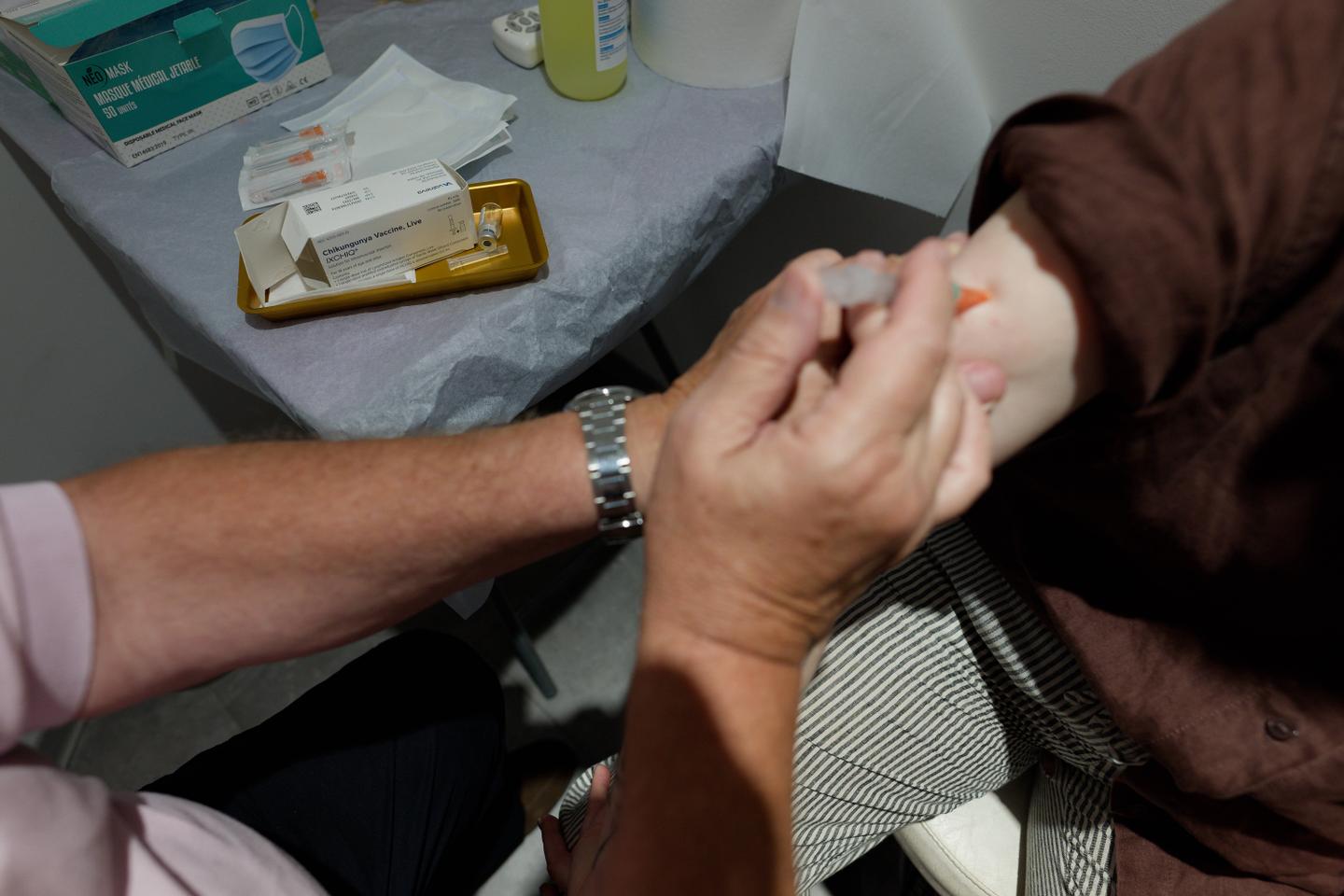
US health authorities have suspended the licence for the Ixchiq vaccine against the chikungunya virus following reports of “serious adverse events”, the drug’s French maker said on Monday.
Ixchiq is one of just two vaccines approved by the US Food and Drug Administration (FDA) for the mosquito-spread virus, which mainly occurs in tropical and subtropical regions but has recently been discovered in countries worldwide.
French company Valneva obtained US approval for the vaccine in 2023, but reports of side effects have prompted reviews, in particular over its use in older patients, including by the European Medicines Agency this year.
Your personal data will be processed and information from your device (cookies, unique identifiers, and other device data) may be stored by, accessed by and shared with 88 TCF vendor(s) and 20 ad partner(s), or used specifically by this site or app.