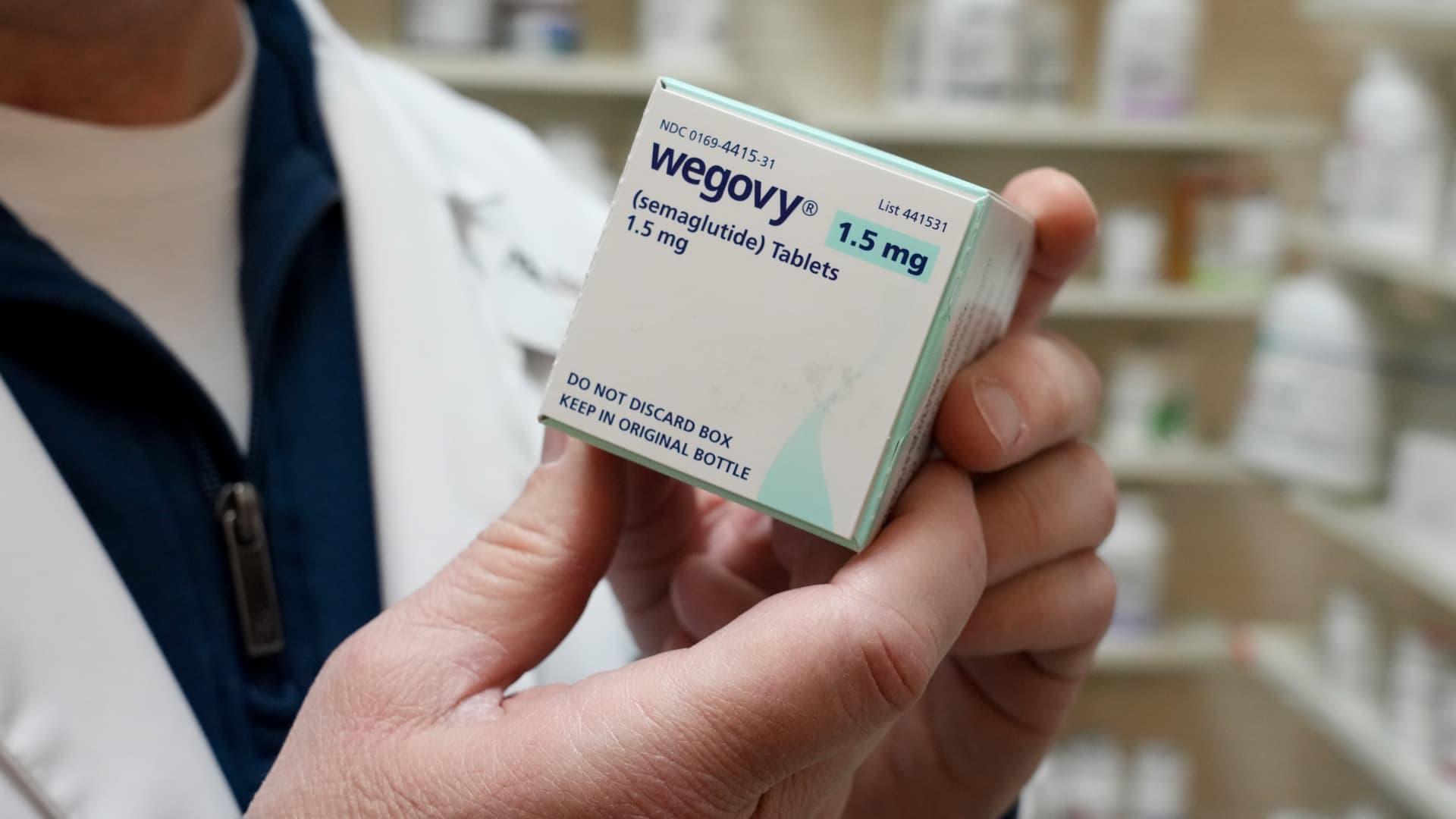
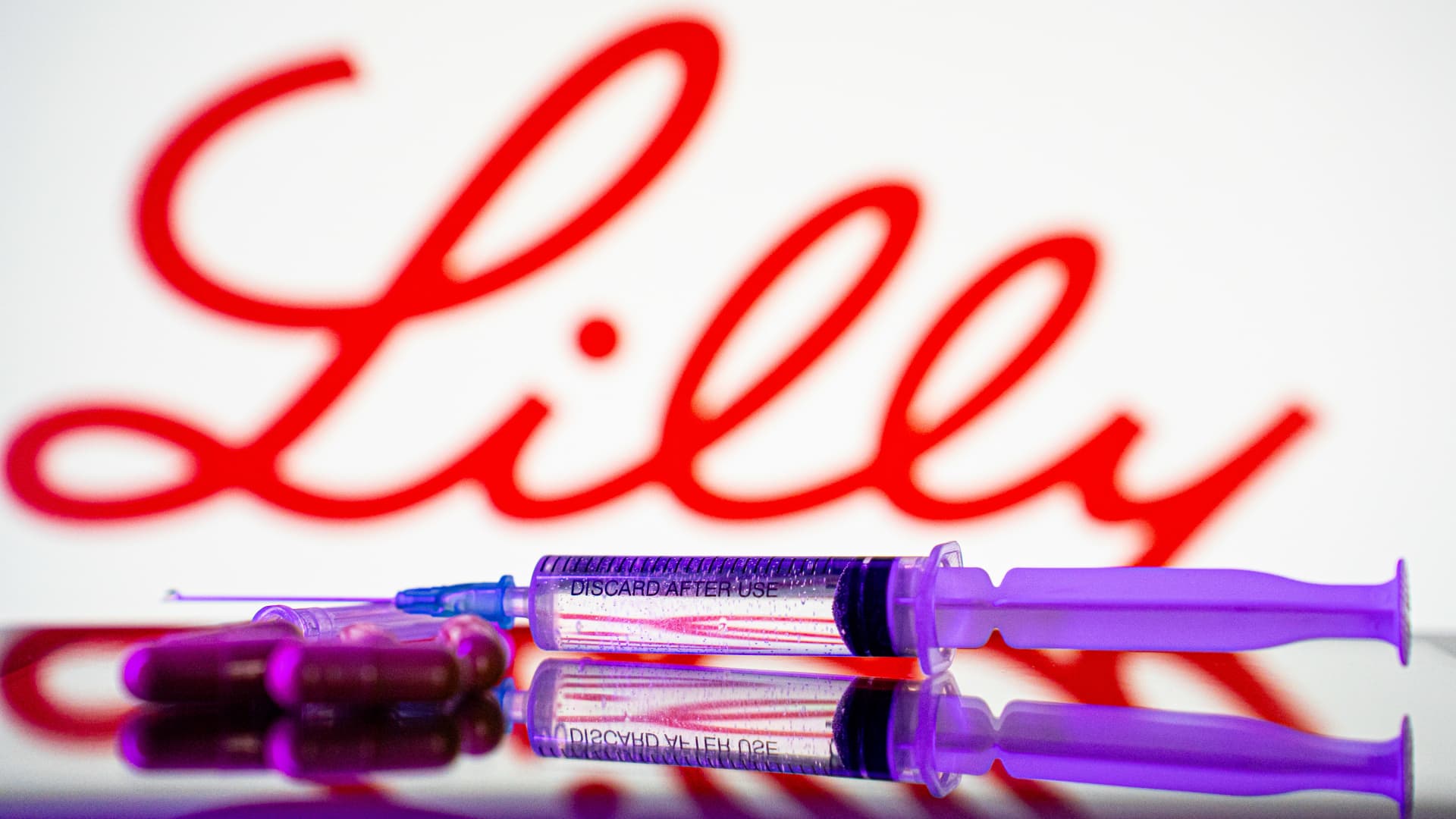
Samsung Epis Holdings, Daewoong, Dongkook, Yuhan race to develop long-acting formulations through advanced drug delivery platforms (123rf) With Eli Lilly’s diabetes and obesity treatment Mounjaro overtaking MSD’s cancer therapy Keytruda as the world’s top-selling drug in the first quarter, Korean drugmakers are focusing on developing monthly obesity shots that involve less frequent injections.Mounjaro and Novo Nordisk’s diabetes and obesity treatment Wegovy currently require weekly injections.Samsung Epis Holdings’ two subsidiaries — biopharmaceutical developer Samsung Bioepis and biotechnology platform developer Epis NexLab — teamed up with local biotech G2GBio to develop long-acting obesity treatments by leveraging the latter’s microsphere-based drug delivery technology in March.G2GBio features long-acting injection technology that can extend the drug efficacy to months with major pipelines of semaglutide-based drug candidate. Semaglutide is a glucagon-like peptide-1, or GLP-1, receptor agonist that mimics a natural hormone to control blood sugar, decrease appetite and slow gastric emptying. Wegovy is based on semaglutide.Daewoong Pharmaceutical announced a strategic partnership with local bio startup TionLab Therapeutics to co-develop long-acting monthly semaglutide-based injectable last week. According to the announcement, the collaboration will combine TionLab’s platform Quject Sphere and Daewoong’s platform CURE.The companies said Quject Sphere will control the speed of early release while CURE will maintain a stable release rate for the desired period thereafter with the goal of cutting down the number of injections from 52 times a year to 12 times a year.Daewoong, which is leading the clinical studies for the partnership’s long-acting weight-loss treatment, submitted an investigational new drug application to the Korean authorities last month, looking to begin the trial before the end of the year.Dongkook Pharmaceutical already began developing three types of semaglutide-based injections that require a shot every one, two and three months in 2024. Through non-clinical studies, the drugmaker confirmed that its microsphere technology was able to suppress the initial burst while releasing the drug in a stable, continuous manner within the effective concentration range for two months. The company has set eyes on entering Phase 1 clinical study of the long-acting obesity drug next year.Yuhan joined hands with Inventage Lab in 2024 to leverage the latter’s long-acting drug delivery platform technology to develop monthly injections based on both semaglutide and Mounjaro’s main ingredient tirzepatide. Yuhan plans to submit the invetigative new drug applications for both of them this year.“Given the growth forecast of the weight-loss drug market, global pharmaceutical powerhouses that have fallen behind in the obesity drug race are ramping up efforts to pick up the pace,” said an official at a local drugmaker. “Korean companies may look to carry out clinical trials not only in the country but also abroad.”According to Morgan Stanley Research’s latest report published earlier this month, the global market for obesity drug could reach $150 billion at its peak in 2035, a significant increase from the market analysis’ previous forecast of $105 billion made about two years ago. The report said the global weight-loss drug market reported approximately $15 billion in sales last year.
Beyond Mounjaro: Korean drugmakers chase monthly obesity shot
With Eli Lilly’s diabetes and obesity treatment Mounjaro overtaking MSD’s cancer therapy Keytruda as the world’s top-selling drug in the first quarter, Korean d