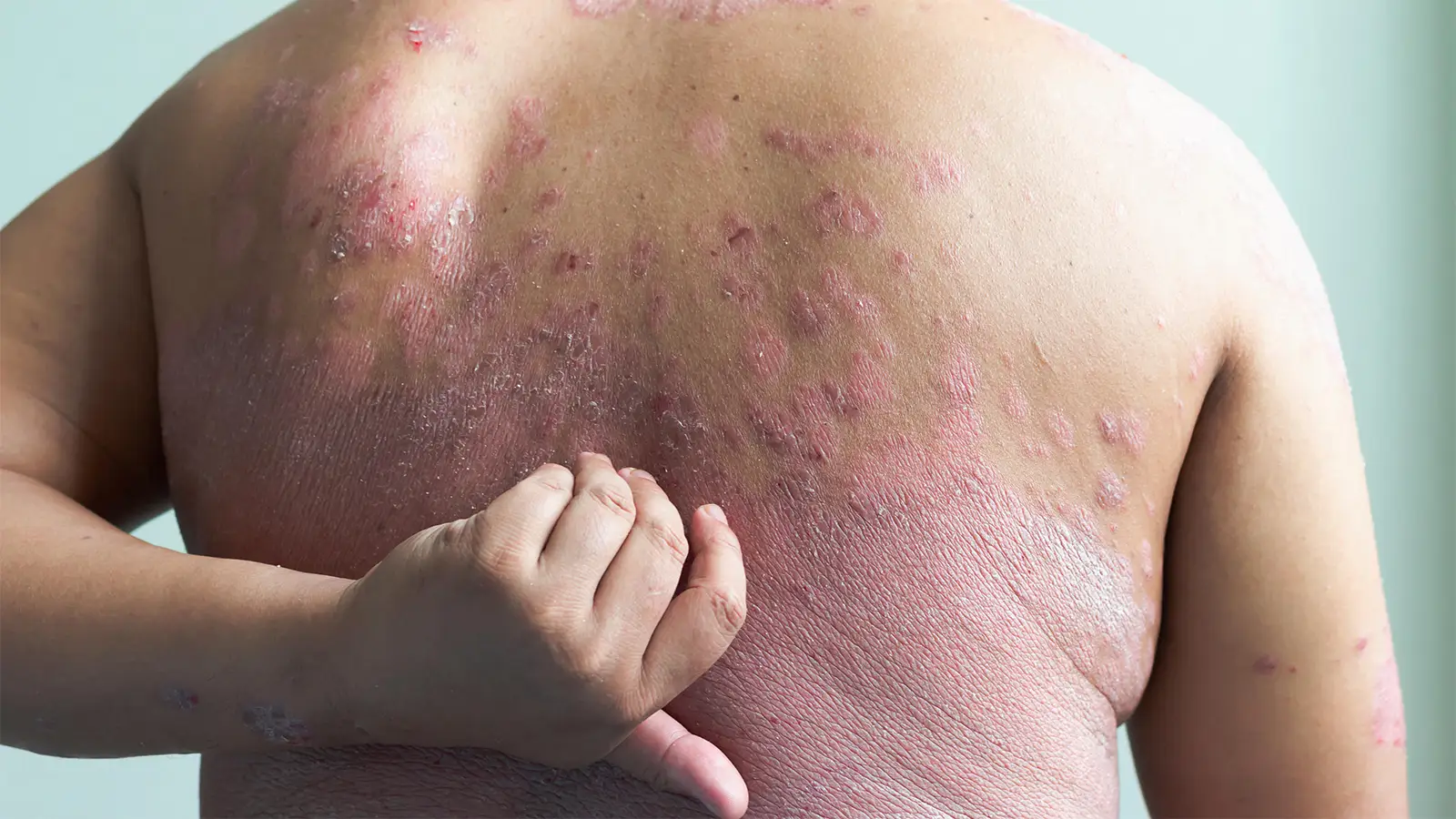
The association between psoriasis and obesity is well established.In this randomized trial, patients with plaque psoriasis and overweight or obesity had significantly better outcomes in terms of skin clearance and weight loss with ixekizumab plus tirzepatide versus the psoriasis drug ixekizumab alone.The results may offer a new approach for the dual management of obesity and psoriasis.
Adding a dual GLP-1/GIP receptor agonist to the psoriasis drug ixekizumab (Taltz) significantly improved outcomes in adults with difficult-to-treat plaque psoriasis due to overweight or obesity, the open-label TOGETHER-PsO trial indicated.
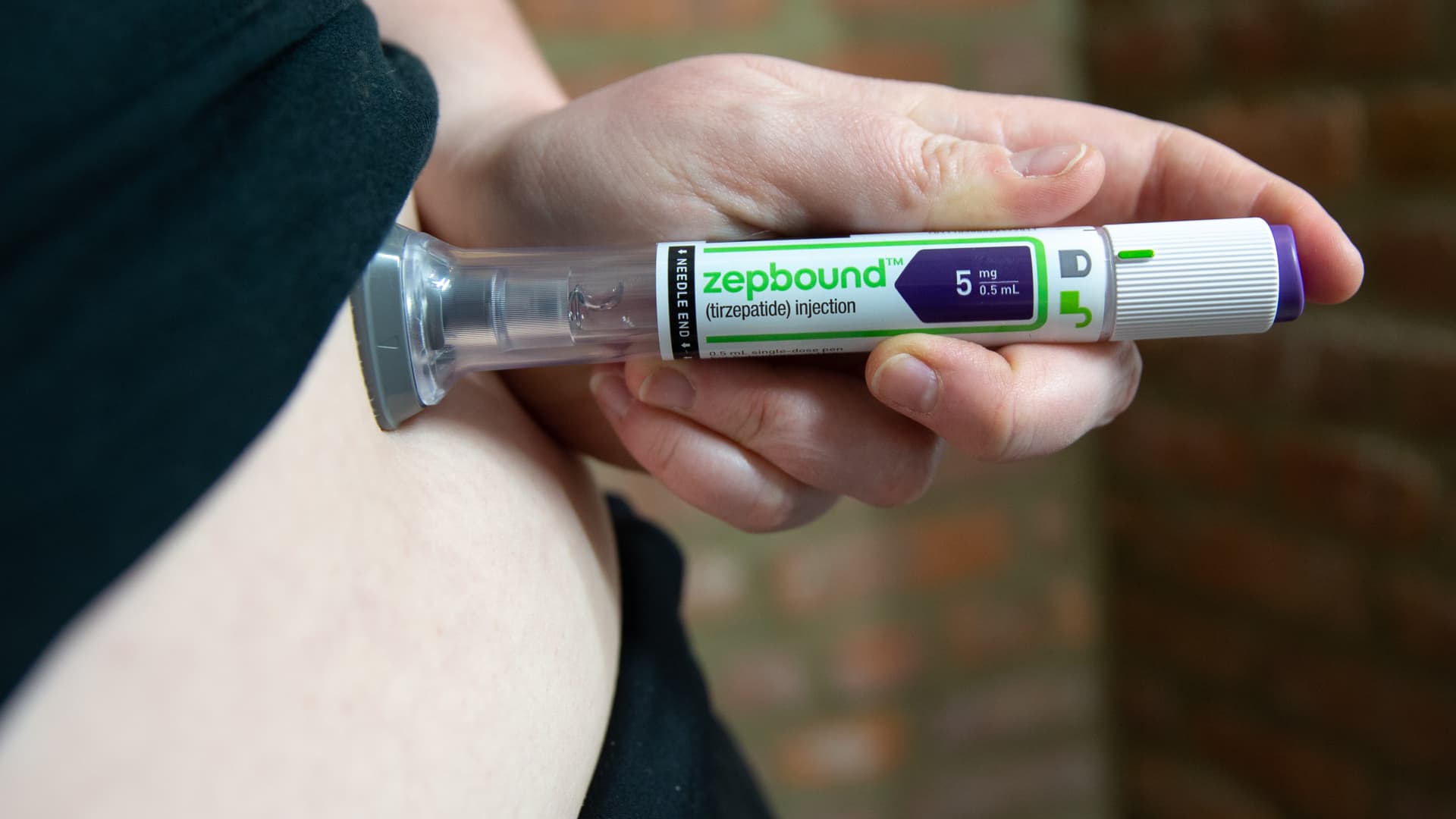
At 36 weeks in the phase IIIb study, 27.1% of patients randomized to ixekizumab plus tirzepatide (Zepbound) achieved the primary endpoint -- a composite of complete skin clearance and at least a 10% reduction in weight -- as compared with 5.8% of patients assigned to ixekizumab alone (P<0.001), reported researchers led by Mark Lebwohl, MD, of the Icahn School of Medicine at Mount Sinai in New York City.
More patients assigned to tirzepatide also met criteria for complete skin clearance alone (Psoriasis Area Severity Index [PASI] 100), with respective rates of 40.6% and 29% (P=0.04). Further benefits were seen in skin-related quality of life and in triglycerides and blood pressure as well, according to findings published in JAMA Dermatology and presented at the Society for Investigative Dermatology annual meeting in Chicago.