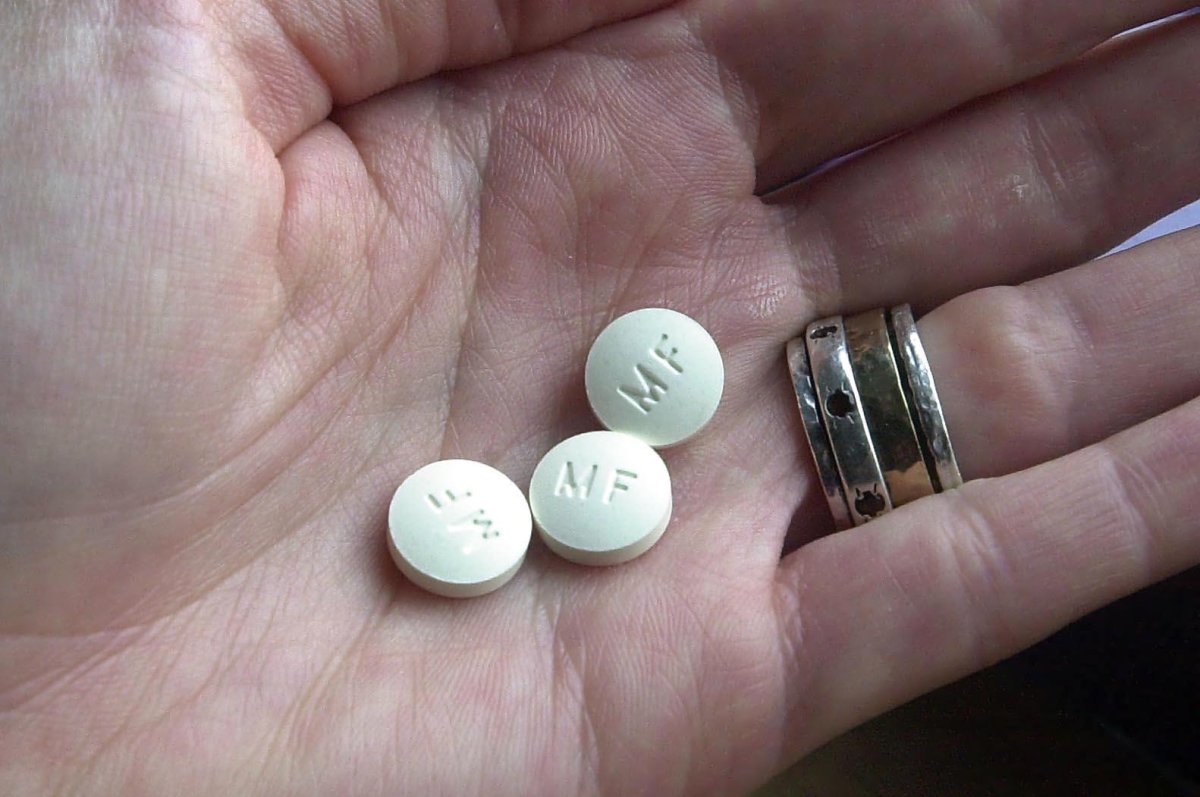
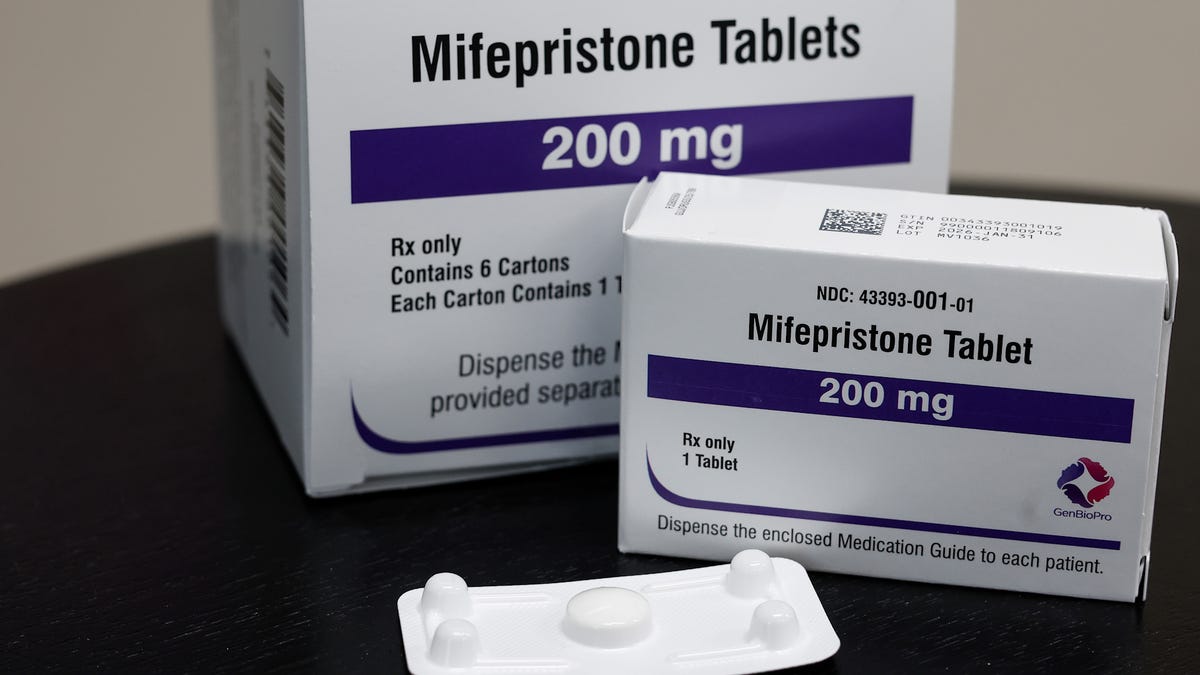
WASHINGTON – Two years after the Supreme Court tossed out a challenge to the widely used abortion drug mifepristone, the justices must again decide whether access should be restricted.
In an emergency filing on May 2, mifepristone maker Danco Laboratories asked the court to immediately pause a lower court ruling limiting access.
The conservative 5th U.S. Circuit Court of Appeals on May 1 temporarily reinstated a Food and Drug Administration requirement that doctors prescribe the drug only after an in-person exam. The three-judge panel did so in response to a challenge from Louisiana to the elimination of that requirement by the Biden administration.
More: Court blocks access through the mail to mifepristone abortion pill
Louisiana argues that allowing the drug to be dispensed through the mail ignores the threat of complications from mifepristone, such as sepsis and hemorrhaging, The state also says mailed delivery of the drug allows women to get around abortion bans.