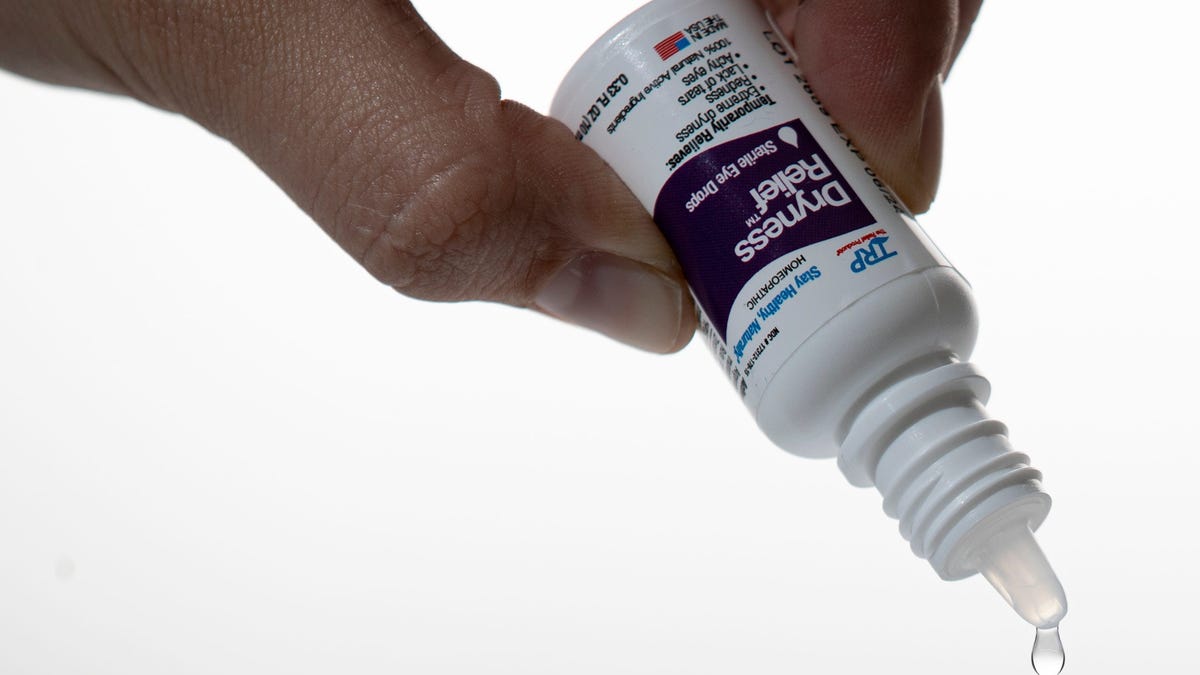
Millions of eye drop products have been recalled over concerns they may not be sterile, according to an enforcement report from the U.S. Food and Drug Administration.
On March 3, eye care product distributor K.C. Pharmaceuticals issued a voluntary recall for more than three million bottles of eye drops.
The eight impacted product batches were sold under various brand names and have expiration dates ranging from April to October. They were distributed to nationwide retailers like Walgreens, Kroger and CVS.
The FDA categorized it as a Class II recall on March 31. Class II, the agency's second-highest recall class, indicates that using the impacted product could cause temporary adverse health consequences.
USA TODAY has reached out to K.C. Pharmaceuticals.