In this article
The U.S. Food and Drug Administration approved Eli Lilly
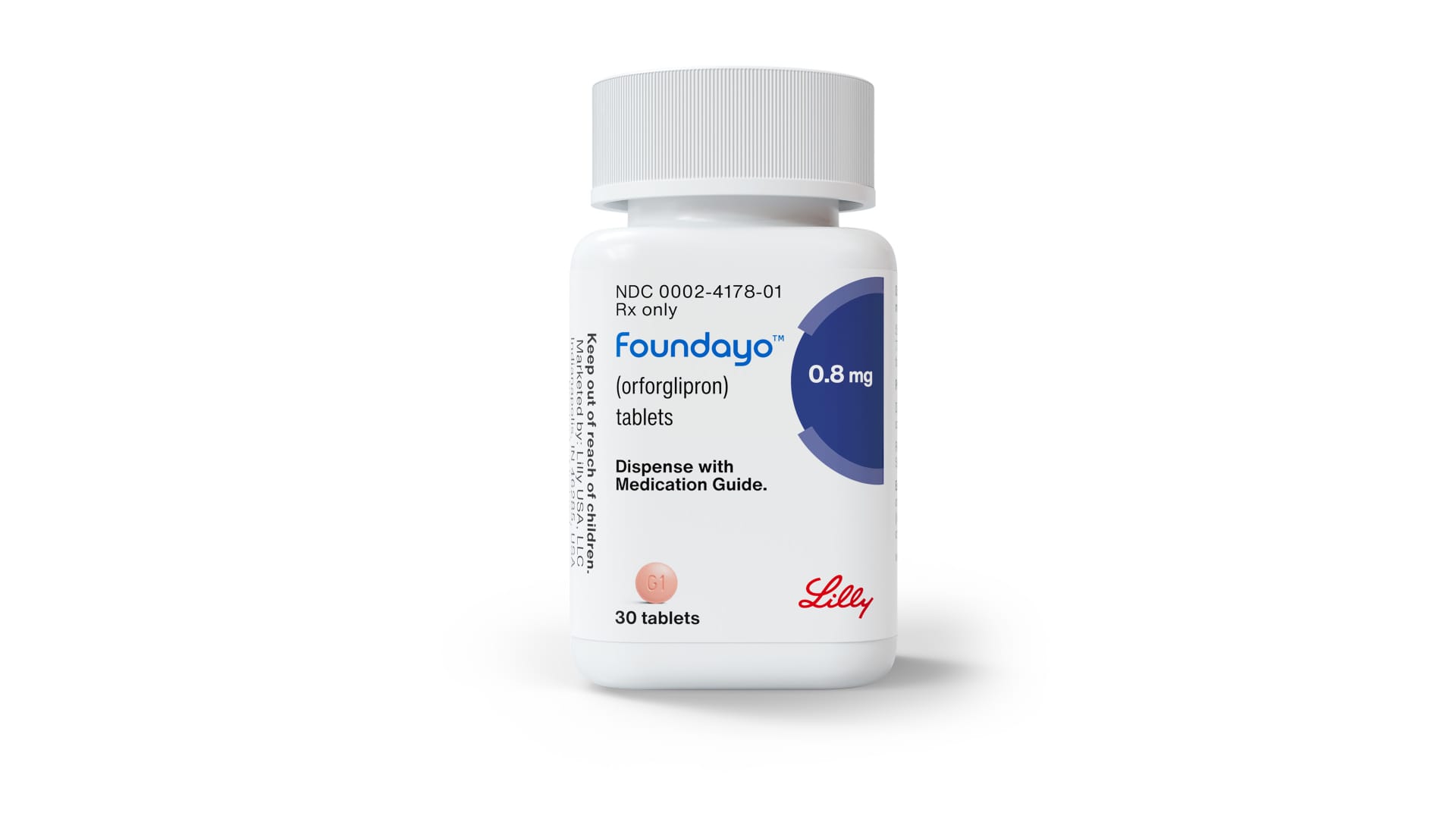
’s GLP-1 pill, the company said, a major milestone for the Indianapolis-based drugmaker and one that will test the market for new weight-loss medications.
The approval comes just a few months after Lilly submitted the drug to the FDA as part of a program that grants speedy reviews for drugs that are considered national priority interests. That means Lilly will introduce its Foundayo only about three months behind Novo Nordisk’s Wegovy pill, setting the stage for the next battle between the rival drugmakers in the next frontier for GLP-1 drugs.
“It’s a big moment,” Eli Lilly CEO Dave Ricks said in an interview with CNBC. “We’ve obviously been working in this category of medicines for a while with the first GLP-1 medication 20 years ago and improving ever since. Here is an option that’s not more effective ... but it’s more accessible, it’s easier to fit into your daily routine.”