In this article
Johnson & Johnson
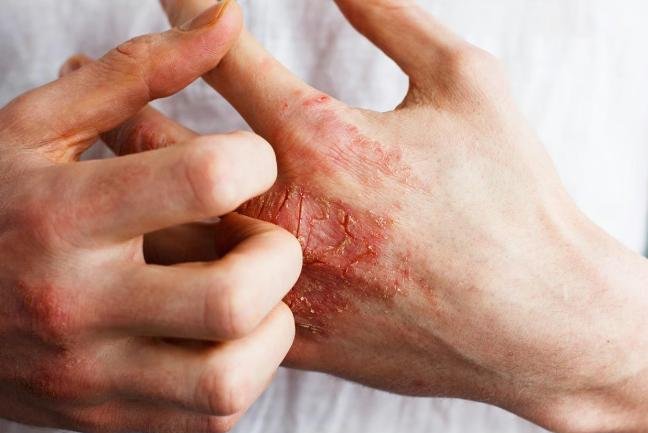
said on Friday it discontinued a mid-stage study of its experimental drug to treat patients with moderate to severe atopic dermatitis after it failed to meet efficacy goals in an interim analysis.
The drug, JNJ-5939, was being tested in patients with atopic dermatitis, or eczema, a skin condition that causes inflammation, redness and intense itching.
The drug was well-tolerated but did not meet the “high-bar” efficacy required to advance development, the company said.