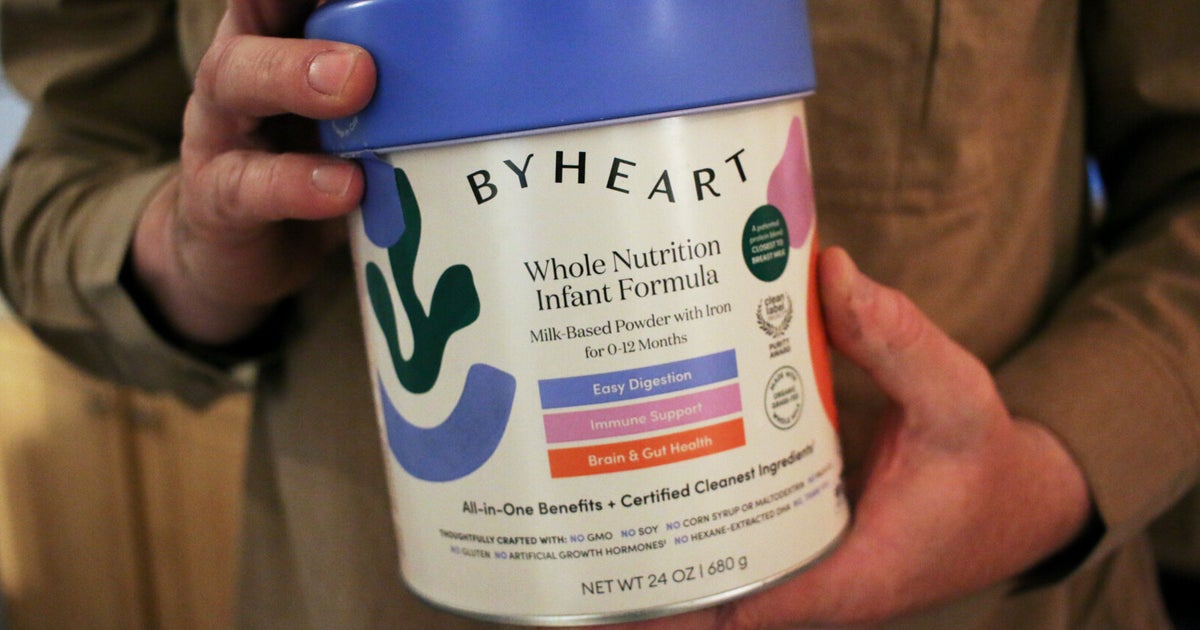
Four of the nation’s top retail stores failed to promptly pull contaminated infant formula tied to a dangerous botulism outbreak from their shelves, federal health officials said in warning letters posted Monday.
The U.S. Food and Drug Administration sent letters to leaders at Walmart, Target, Kroger and Albertsons, saying the companies continued to sell ByHeart infant formula for days or weeks, despite a Nov. 11 recall of all products in the outbreak that has sickened more than 50 babies in 19 states.
“As a participant in the supply chain, your firm should take prompt and effective action when notified of a product recall,” FDA officials said in warning letters sent to the companies on Dec. 12 and posted online Monday.
The formula was found at Target stores in 20 states “well after the recall was initiated,” one letter said. In addition, it was sold at a Target store in New Hampshire on Nov. 16, despite an electronic block on the product’s sales code, the FDA noted. And at a Target store in Arkansas, single-serve packs of ByHeart formula were promoted with a “Sale!” sign and a $2 discount from Nov. 16 to Nov. 22.
Information from state and local health officials said ByHeart formula was found at Walmart stores in 21 states from Nov. 12 to Nov. 26. The formula was found in Albertsons stores in 11 states from Nov. 12 to Nov. 19, and at Kroger stores in 10 states from Nov. 12 to Nov. 19.