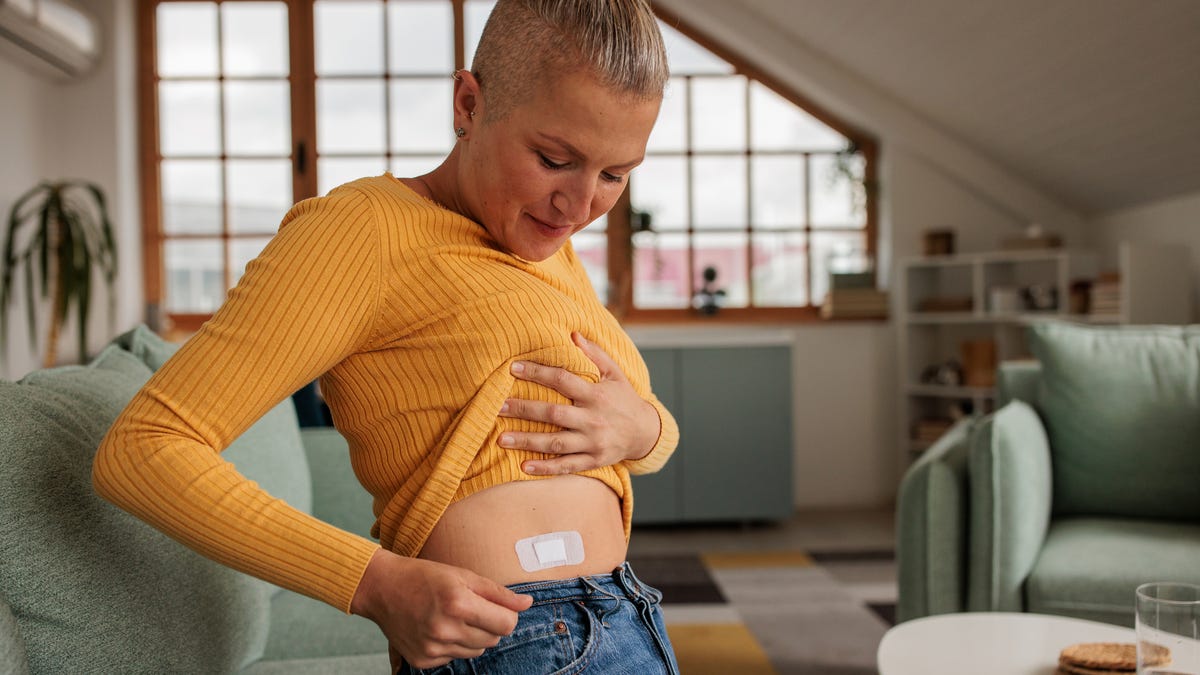
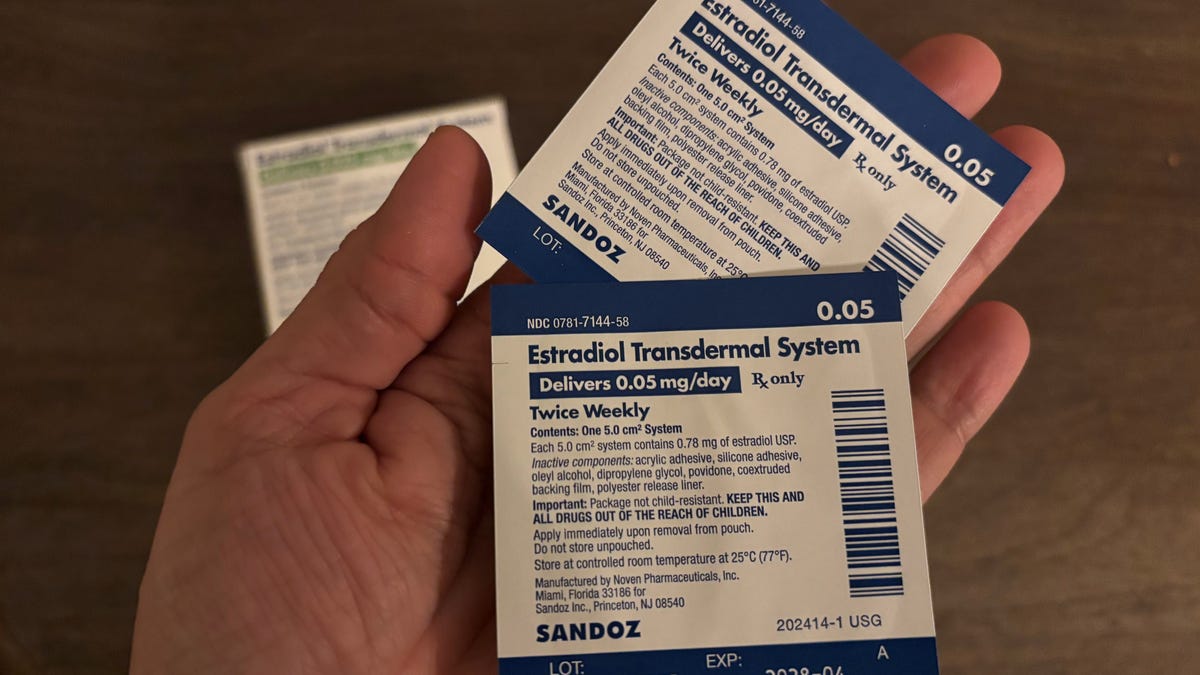
For the past decade, Dr. Heather Hirsch has tried to convince women that hormone therapy drugs are the best way to treat symptoms of menopause and perimenopause. But warning labels on estrogen have scared many women away from getting the care they need.
Now the Food and Drug Administration is asking drug companies to remove these labels, known as black box warnings, on hormone therapy drugs containing estrogen. This change will be monumental in helping more women manage disruptive menopause and perimenopause symptoms ranging from increased urinary tract infections to night sweats.
“The label was outdated and harmful,” says Hirsch, a physician and founder of the Menopause Clinic at Brigham and Women’s who testified in front of the FDA to remove the labels this summer. “It led to so many women not getting the care they need.”
A 2002 Women’s Health Initiative (WHI) study linked hormone therapy to slightly higher risks of breast cancer, heart attacks and strokes in postmenopausal women. The risks, recognized later, were mostly found in women who were older when they started hormone therapy.
The ramifications from the study affected millions of women.