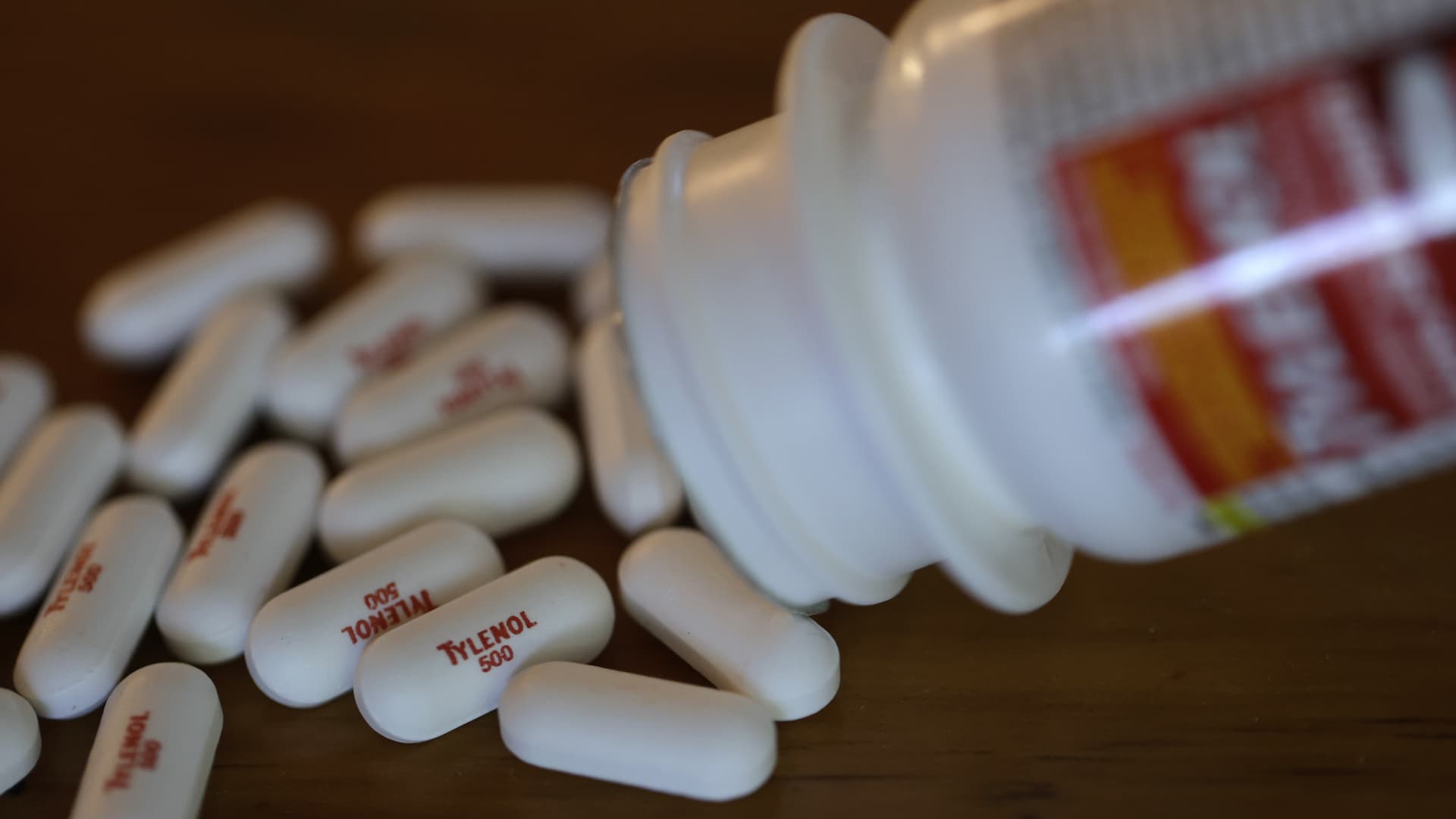
The maker of Tylenol is urging U.S. health regulators not to add an autism warning label to it and other pain-relievers containing acetaminophen, calling the request "unsupported by the scientific evidence and legally and procedurally improper."
Kenvue's response, dated Oct. 17, came after a citizen petition sought new pregnancy warnings for acetaminophen.
The petition, filed by the Informed Consent Action Network, asked the U.S. Food and Drug Administration to note a possible link between prenatal exposure and autism or attention-deficit/hyperactivity disorder.
The petition piggybacked on recent remarks by President Donald Trump, who said Tylenol might cause autism -- a claim many health experts say is not supported by science.
"For over a decade -- and as recently as August -- FDA has fully evaluated the emerging scientific evidence and repeatedly concluded that the data do not support a causal association between acetaminophen use in pregnancy and neurodevelopmental disorders such as autism," Kenvue said.