ByThomas Brewster,
Forbes Staff.
S
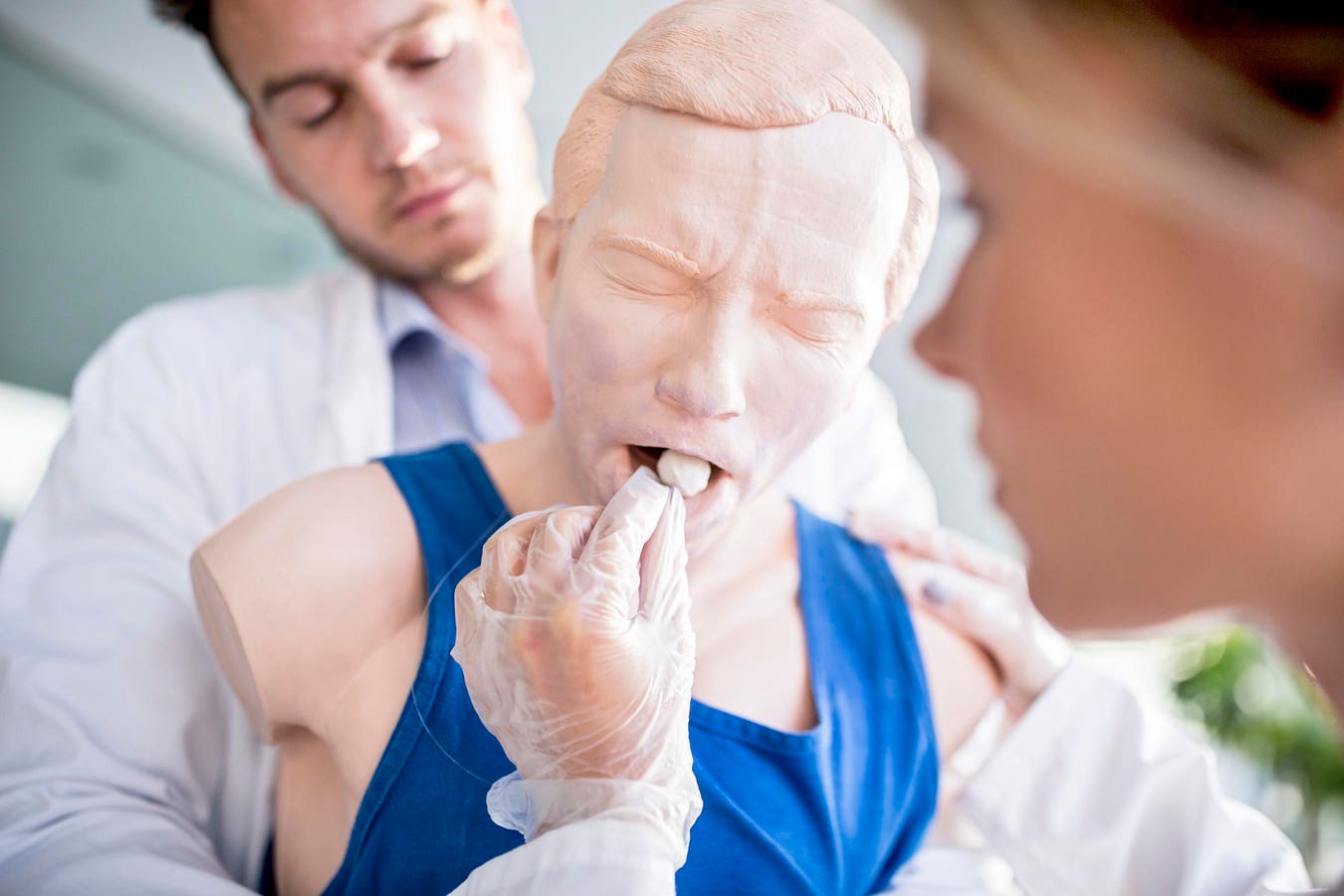
ince founding their company 15 years ago, Colorado-based Alan Carver and his wife have sold tens of thousands of what they called “Dechokers,” a device that’s like a plunger for humans that claims to be able to remove items blocking airways and stop people from choking to death. Marketed primarily as an FDA-approved device for use on infants and the elderly, the Dechoker was sold on Amazon and on its own website. On the Dechoker website, it explicitly stated, “the FDA has helped Dechoker with our processes and we consider their input a vital piece to Dechoker's future success.”
But the FDA had not only never approved the Dechoker, it had told the Carvers to stop selling the product entirely in 2023, according to a seizure warrant for the founders’ funds. It should have been categorized as a Class III medical device, the highest risk bracket, because, as with other over-the-counter anti-choking devices, it hadn’t been checked for safety, the agency told them. That allegedly didn’t stop the Carvers, who continued to sell the Dechoker on Amazon, raking in $8.2 million in sales between 2023 and May 2025, per the warrant, which was unsealed earlier this year.