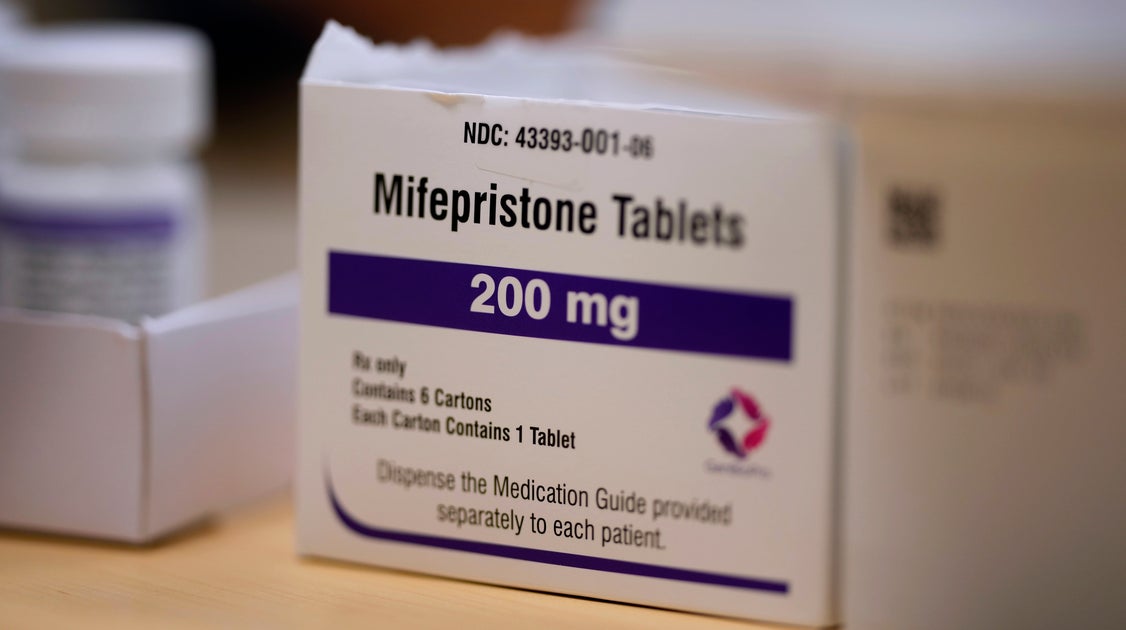
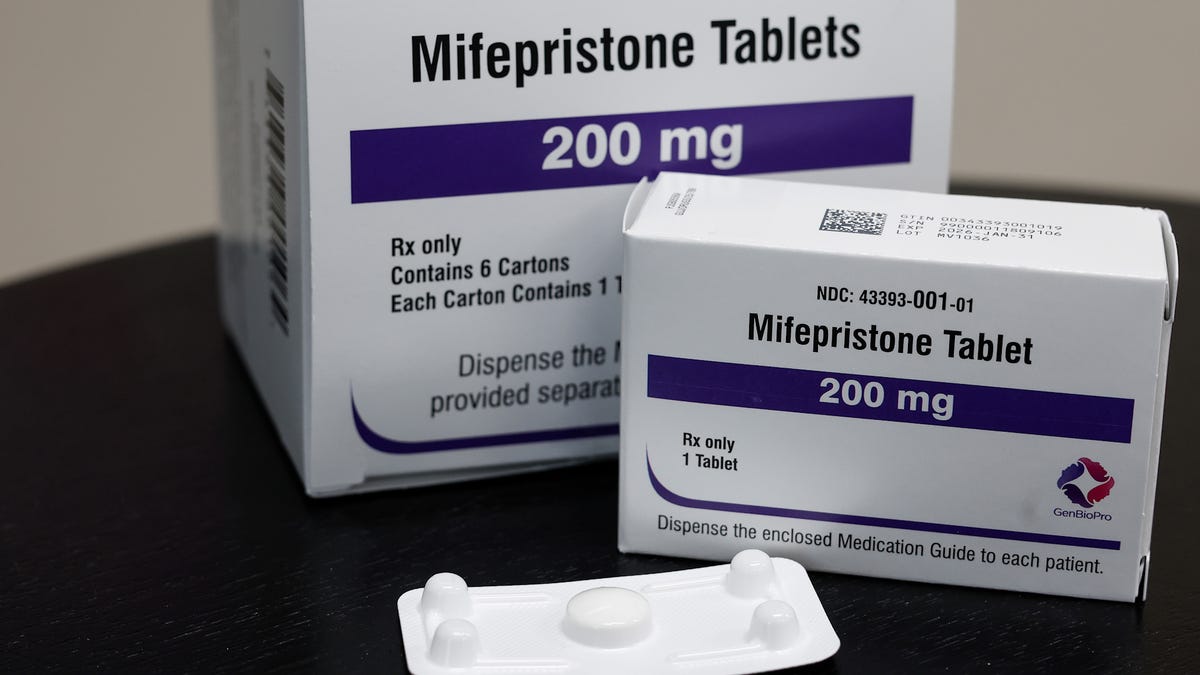
The Food and Drug Administration has approved a new generic version of the abortion pill mifepristone, prompting an outcry from conservatives who’ve been eager to restrict access to the medication.
The generic pill was approved in September, according to Evita Solutions, its manufacturer. It joins another generic pill that’s already on the market from GenBioPro, which was approved in 2019.
Andrew Dixon, a spokesperson for the Health and Human Services Department, noted that the FDA “has very limited discretion in deciding whether to approve a generic drug. By law, the Secretary of Health and Human Services must approve an application if it demonstrates that the generic drug is identical to the brand-name drug.”
Dixon added that the FDA “does not endorse any drug product and directs prescribers to follow all labelling.”
The FDA’s approval of the medication greenlights another low-cost way to obtain the abortion pill, which has seen a major uptick in usage since the fall of Roe v. Wade, a decision that paved the way for numerous states to impose severe restrictions on reproductive rights.