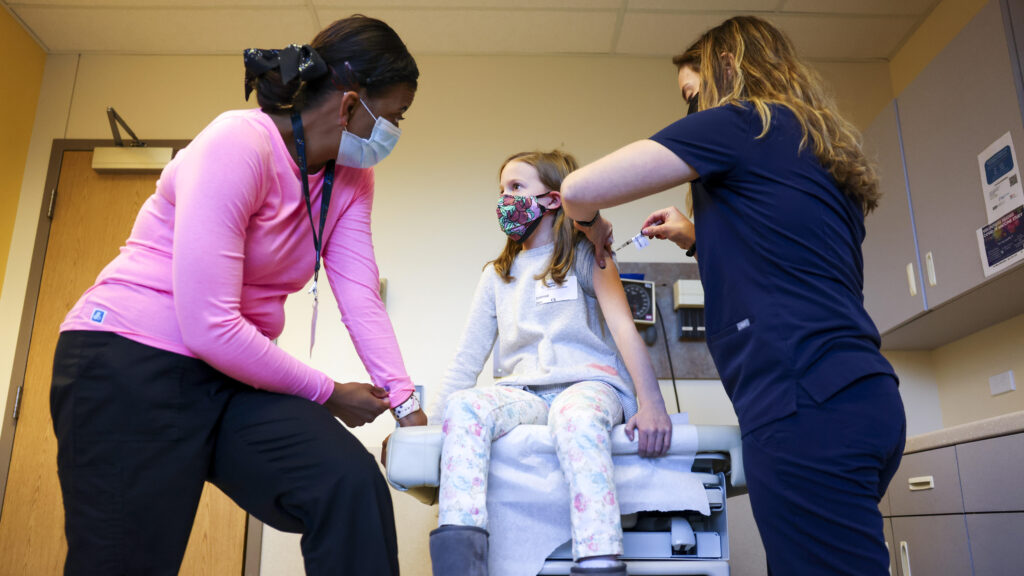
May 18 (UPI) -- The U.S. Food and Drug Administration approved Novavax's Covid-19 vaccine with age restrictions after a six-week delay.
The agency approved the vaccine only for people 65 and older and those 12 and older with at least one underlying condition in which they are at a higher risk of severe illness. The company is based in Gaithersburg, Md.
The conditions include older age, asthma, diabetes, lung disease, obesity and pregnancy.
"Market research and US C.D.C. statistics indicate that older individuals and those with underlying conditions are the populations most likely to seek out COVID-19 vaccination seasonally," Novavax President and CEO John Jacobs said in a statement Saturday to CNN. "This significant milestone demonstrates our commitment to these populations and is a significant step towards availability of our protein-based vaccine option."
Related